Artist Studio
By Helen Herlocker
ChemMatters, April 2012
- Download article
- Teacher’s Guide
Word | PDF
In Frog Valley, W.Va., Mark Schwenk, a metal artist and photographer, has been building and operating a pottery kiln for the past 3 years. Visitors enjoy going to his studio because they are in for a show! Schwenk opens the kiln, grabs a red-glowing pot with metal tongs, and plunges it into a metal trash can stuffed with shredded paper. Flames shoot skyward! Then, Schwenk slams the lid down, and the flames are replaced by a small emission of black smoke. After a few minutes, Schwenk removes the pot, still very hot, and dips it in a tub of water, where the billowing steam captures the attention of a crowd of visitors. When the pottery piece is cleaned, it has a beautiful shiny and colorful glaze.
Schwenk makes Raku pottery, an increasingly popular art form that is also a fun way to fire pottery. He is one of seven artisans working together as part of an artist collective called Frog Valley Studios. The other artisans create craft from metal, ceramics, and glass, which attract many visitors throughout the year.
I work at Frog Valley Studios as a volunteer apprentice. I help Schwenk and a stained-glass artist named Veronica Wilson with pottery and stained glass. I find Raku pottery and stained glass fascinating. Let me show you around!
Red-hot pottery
It’s hard to imagine a craft as basic as pottery, which consists of making pots out of clay—a naturally occurring material composed mainly of fine-grained minerals. Essentially, pottery consists of sculpting pots by hand, decorating them with a glaze, and heating them to high temperatures in a kiln so they can harden before letting them cool down outside.
But unlike traditional pottery, Raku pottery is more exciting. Sculpting a Raku pottery piece and glazing it is similar to traditional pottery, but unlike the gradual heating and gradual cooling of the everyday pottery kiln—a process that takes at least 8 hours—a Raku kiln is best described as “fast and furious.” The pots sit in a chamber, exposed to a fuel-rich flame that rapidly heats them to 1,800 oF.
What Schwenk shows his visitors a few times per year is the most spectacular part of the process, but also—according to many—the most fun and enjoyable. This unusual form dates back to the mid-16th century when Sen no Rikyu, a Japanese tea master and former Zen monk started firing pieces in such a way that each bowl presented its own unique shape, texture, and surface features. Today, Raku potters produce pieces which, like those of Sen no Rikyu, have singular forms and features. In short, every piece is a surprise!

What happens in the trash can is an example of oxidation-reduction, or redox, reactions—among the most common and most important chemical reactions in everyday life. A redox reaction involves both oxidation (loss of electrons) and reduction (gain of electrons). In this case, the elements are the metals in the glaze, which either lose or gain electrons. The elements that lose electrons are “oxidized” and those that gain electrons are “reduced.”
The glazes contain metal of either the oxide ion (O2−)—such as copper(I) oxide (Cu2O) and silver oxide (Ag2O)—or the carbonate ion (CO32–), such as cobalt(II) carbonate (CoCO3) and copper(II) carbonate (CuCO3).
The burning in the trash can occurs due to the presence of three elements: heat, fuel, and oxygen. With the lid cutting off the supply of atmospheric oxygen, the nearest available source of oxygen is the glaze compounds, which all contain oxygen.
The metals in the glaze components are reduced, while the carbon or carbon monoxide coming from the incomplete combustion is oxidized. For example, in the case of copper oxide (Cu2O), which is actually a crystal composed of the ions Cu+ and O2–, the following redox reactions occur in the can:
Reduction: The copper ions are reduced into elemental copper:
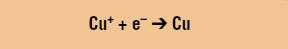
Oxidation: The carbon is oxidized into carbon dioxide in the presence of carbon or carbon monoxide:
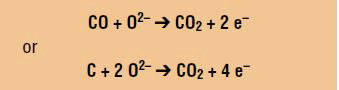
The combination of both reduction and oxidation reactions leads to either of the following reactions:
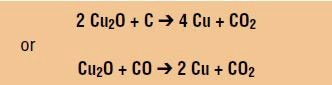
The end result is a shiny copper coating on the surface of the pot that replaces the copper oxide that was present in the glaze before the combustion occurred.
What makes Raku pots unique is the presence of different metallic streaks that would not form if the pots were left outside to cool down after being taken out of the kiln.



Art on glass
Another art form that is gaining popularity is stained glass. To picture what stained glass looks like, imagine a beautiful painting that you like. Now imagine the painting with sunlight streaming through. That’s essentially what a stained-glass window looks like. It consists of small pieces of glass that are carefully shaped and assembled to form a design or a picture. Stained glass is also used to make lamps, boxes, and decorations, and anyone can learn this craft.
At Frog Valley, stained-glass artist Veronica Wilson created a stained-glass window featuring Appalachian wildlife—frogs, birds, luna moths, turtles, and a brightly coiled snake. In the sunlight, the stained-glass window looks like a brilliant array of colored glass pieces—some smaller than your little finger nail—held in place by a metallic support network.
Wilson explains that her work, with its small scale and tiny mosaic pieces, is actually a scaled-down version of the stained-glass windows that grace cathedrals and churches. To create such windows, artists connected large window pieces with a reinforcing material, called a lead came (Fig. 1), which was soldered at the junctions between the window pieces.
Wilson uses bits of colored glass to form small flower petals, insect wings, stems, and leaves. But instead of using lead cames, she fastens them in place with a different technique. First, she draws the shapes on paper, glues them lightly on a piece of colored glass, and then cuts around them (Fig. 2).

Using a glass-cutting tool, which she holds like a vertical pencil, she scores the surface around each shape. Then, she lightly taps on the underside of the glass to weaken the scored line and applies a downward pressure with glass pliers on each side of the scored line to make the break. When the small pieces are assembled together, the work looks like a colorful jigsaw puzzle.
To put the small pieces together, Wilson uses copper foil, a spool of lead-tin solder, and a cleaning agent. First, Wilson edge-wraps each piece of glass with a narrow strip of copper foil—today’s foil is available in spools with a backing of adhesive—which she attaches to the glass (Fig. 3).

Hydrochloric acid removes any oxides that are present on the surface of the copper foil, so that only copper is exposed to the solder:

The product, copper(II) chloride (CuCl2), is soluble in water and easily washes away. What is left is a surface of pure copper, to which the tin-lead solder can bind.
The solder is a mixture of metals with low melting points. Wilson uses a tin-lead 50:50 mixture. She heats the copper base and moves the hot iron to melt a bead of solder into place (Fig. 4). When the tin-lead metal cools down, it adheres to the copper and holds the glass in place.

Wilson then applies a thin shiny coating called a liquid patina to give the desired color to the solder line and to protect the cooled solder from further oxidation. Days, sometimes weeks, in the making, the piece is finally ready to be metal-framed and mounted in a sunny window where it blooms to life!
Want to give it a try?
After seeing the art pieces at Frog Valley Studios, I wanted to become familiar with how stained glass and Raku pottery are created. So, I simply showed up and volunteered to help the artists with the many steps involved in the production. Hours of free help is hard to turn down! And they became valuable and fun lessons.
You may want to try Raku pottery or stained glass. Classes are offered throughout the United States, and chances are, classes are available near you. While you learn about the properties of the materials and how they interact with each other, you will discover that it really is all about chemistry!
Selected references
- Baxter, R. Glass: An Amorphous Solid. ChemMatters, Oct 1998, pp 10–11.
- Stone, C. The Terra Cotta Warriors: Army from the Earth. ChemMatters, Feb 2000, pp 14–15.
- Rohrig, B. Myths: Chemistry Tells the Truth. ChemMatters, Dec 2010, pp 8–10.
Helen Herlocker, a science writer who makes pottery and stained glass near Berkeley Springs, W.Va., is a former managing editor of ChemMatters. Her latest ChemMatters article, “Retiring Old Tires” (coauthored with Donald Jones), appeared in the April 2007 issue.
Also in this Issue (April 2012)

Lighten Up!
What are the advantages and potential drawbacks of compact fluorescent light bulbs over regular light bulbs?

Recycling Aluminum: A Way of Life or a Lifestyle?
In the Western world, recycling aluminum helps to protect the environment. In India, it is a means of livelihood for thousands of people. Learn how recycling aluminum is changing lives and helping the environment.

