The Dirt on Getting Clean
By Barbara Sitzman and Regis Goode
ChemMatters December 2011
- Download article
- Teacher’s Guide
Word | PDF
Regis: Flu season is upon us! Doctors tell us to keep our hands clean to prevent the spread of disease. My students ask what method will best clean their hands.
Barbara: Flu and colds are both caused by viruses. So, the first line of defense is a flu shot. After that there are two general approaches to eliminating viruses from your hands, but each involves complications. One way is using hand sanitizers; the other is just washing with old-fashioned soap and water.
Regis: Soap and water are not always available, but you can easily carry a hand sanitizer. Commercial sanitizers often contain alcohol and other chemicals that effectively kill most viruses. To be effective, the active ingredients should include at least 60% alcohol.
Babara: Be wary of recipes for homemade sanitizers on the Internet. Is this one really a good, inexpensive solution?
Bubble Gum Sanitizer
½ cup aloe vera gel + bubble gum essence
¼ cup of 99% rubbing alcohol isopropanol
Let’s do the math: 99% of ¼ cup of alcohol = .2475 cup of alcohol/.75 cup of solution x 100% = 33% alcohol! While the scent may be appealing, this gel does not contain enough alcohol to kill viruses. Unfortunately, in addition to their effect on viruses, alcohol-based sanitizers kill all bacteria, not just pathogens. Some bacteria are beneficial and actually strengthen our immune systems.
Regis: Also popular is washing with antibacterial soaps. However, this does not kill viruses, the cause of the flu and colds. You may be surprised by the number of products that contain the antibacterial agent triclosan (Fig. 1), a chemical that slows bacterial growth (antibacterial soaps); prevents dental disease (toothpaste and brushes); controls growth of odor-causing bacteria (socks); prevents bacterial degradation (baby pacifiers); and acts as a preservative (cosmetics).
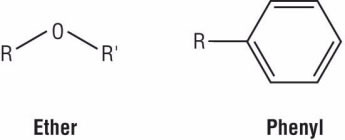
Barbara: Even in low concentrations, triclosan increases thyroid production of hormones that control development of the body, brain, and immune system. When it gets into wastewater, only 4% is removed at the treatment plant, leaving the rest to impact our aquatic environment. In water exposed to sunlight, 1–12% of triclosan converts into extremely toxic dioxins. Note the similarities of the two structures. Both contain ether (oxygen atom bonded between two carbon atoms) and phenyl (benzene ring) functional groups. As predicted by its structure, triclosan is only slightly soluble in water (.012g/L at 20 oC) but is fat-soluble, so it accumulates in human body fat.
Figure 1. Chemical structures of (a) triclosan and (b) dioxin (or 2, 3, 7, 8-tetrachlorodibenzodioxin)
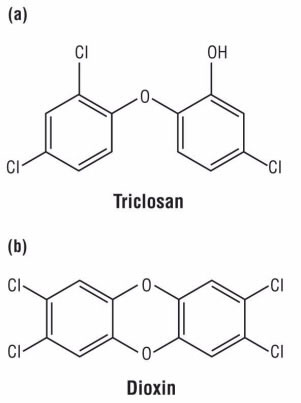
Figure 2. Chemical structures of two functional groups: (a) ether and (b) phenyl.
Regis: Americans overwhelmingly want to use antibacterial products. Have you tried to purchase liquid soap that is not antibacterial? Currently, 75% of liquid soaps on the market are antibacterial, and many contain triclosan.
Barbara: These popular products do not kill flu and cold viruses. Overuse promotes antibiotic resistance in bacteria and encourages viruses to mutate. Just washing and drying your hands removes many viruses. This also reduces disease-causing bacteria which tend to be on the surface, while leaving those that are essential to our well-being.
Regis: While this is true, consider the environmental impact. If you use a paper towel to dry your hands, in effect you are chopping down trees and sending more paper to landfills.
Barbara: The use of antibacterial cleaners also impacts the environment. In a 2008 study by the Centers for Disease Control and Prevention (CDC), triclosan was found in the urine of 75% of the population. The American Medical Association advises against household use, and the Environmental Protection Agency plans a triclosan review in 2013.
Regis: Antibacterial products in hospitals are effective in reducing the spread of many serious infections. The CDC suggests washing their hands for at least 20 seconds with soap or a dime-sized dollop of an alcohol sanitizer for 30 seconds. It is important to let hand sanitizer dry to avoid spreading infection.
Barbara: The needs in hospitals differ from those in the home; should we consider restrictions on home use? The ultimate effectiveness of all soaps, gels, and foams depends on the behavior of the user. Sanitizers are ineffective when hands are soiled or when used by people preparing food. I will reserve hand sanitizers for times when soap and water are unavailable.
Regis: Some decisions are not easy. Should we restrict the use of alcohol-based sanitizers and ban the use of triclosan from over-the-counter products? Will you choose to limit the use of hand sanitizing products in your home? Send us your ideas at: chemmatters@acs.org
Also in this Issue (April 2011)

Mmmm... Flavorful Food!
Taste, smell, color, texture… There are many ways to enjoy food. But explaining why food tastes so great is more difficult because of the variety of flavor chemicals involved. Learn about those chemicals and how they contribute to the flavor of food.

Your Body Under Construction
How do hormones change the bodies and minds of teenagers? We examine the most important hormones and how they work inside the body.
Subscribe to receive full issue!

