Harnessing Solar Power
By Michael Tinnesand
ChemMatters, December 2011
- Download article
- Teacher’s Guide
Word | PDF
People have lived on Utrik Atoll for the past 4,000 years. It is a tropical paradise that is part of the Republic of the Marshall Islands, a nation of islands and atolls in the middle of the Pacific Ocean. Utrik Atoll is a sanctuary for sea turtles, birds, and many species of fish.In recent times, global climate change brought drought to the region, and fresh drinking water was increasingly difficult to come by. What water they could pump from wells relied on electricity from dieselgenerators, but with rising fuel costs, this process was too expensive to sustain.
The Utrik leaders worked with Moana Marine, LLC, a local alternative energy company, to install a solar panel power system and two small wind-powered generators that produced enough electricity to power the island’s water system. Since then, the power system has been providing an abundant supply of electricity to the island’s houses and has allowed the opening of a new school, a new community center, and an agriculture nursery facility.

Utrik may be a model in miniature for the fate that might await the rest of the world in the United States, the main source of energy is fossil fuels —petroleum, natural gas, and coal fossil fuels contribute 85% of the energy that we need to light our homes, power our cars, and cook our meals. But we will eventually run out of fossil fuels. Also, many countries — including the United States — need to import fossil fuels because they are found only in certain parts of the world. Another problem with fossil fuels is that burning them releases carbon dioxide, a greenhouse gas that contributes to global climate change.
A promising alternative to fossil fuels is solar energy. The total human population on earth currently uses about 13 terawatts (1 terawatt equals one trillion watts) of energy. This is only 0.01% of the 120,000 terawatts of energy delivered by the sun to the surface of the earth. If we could only find a way to harness even a fraction of this solar energy, we could solve our energy problems.
Converting sunlight into electricity
To produce electricity from the sun, all that is needed is a small device called a solar cell, which converts solar energy directly into electricity. To power a house or a building, solar cells are combined into modules and arrays to form solar panels (Fig. 1a, 1b).
Figure 1a.
Solar cells are arranged in the form of a module, which are then combined to form an array.

Figure 1b.
A solar panel, such as the one shown here on the roof of a building, consists of a series of arrays that are aligned next to each other.
Solar panels are “first cousins” to the chips inside computers or cell phones. The technology used to make solar panels is similar to making computer chips. Both use a class of material called semiconductors — materials that have a limited ability to conduct an electric current.
Most semiconductors are made from crystalline silicon. In a pure silicon crystal, each silicon atom is bonded to four other silicon atoms, and each bond consists of a pair of shared electrons. This is a stable configuration. Electrons involved in these bonds move very little and are restricted to the bonds.
Silicon does not conduct electricity because its electrons do not move easily. Think about a theater filled with people. If every seat is taken, nobody can move or shift for a better seat. This changes if silicon is “doped.” Doping means intentionally adding a small amount of another element, called a dopant, to silicon.
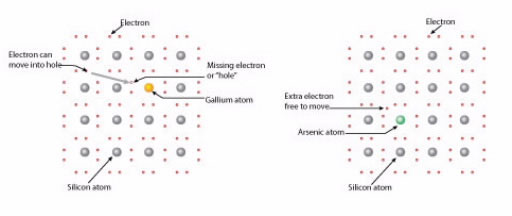
The first kind of doped silicon is called p-type (p is for “positive”). It is produced by introducing atoms — such as boron or gallium — that have one less electron in their outer level than does silicon (Fig. 2a). This means that instead of making four bonds of shared pairs of electrons with other silicon atoms, there is one open “hole.” This is similar to having a few empty seats in a theater; it makes shifting from one seat to another much easier.
The second type of doped silicon is called n-type (n is for “negative”). It is made by including atoms that have one more electron in their outer level than does silicon (Fig. 2b). This additional electron is free to move. In our theater analogy, it is like having one seat occupied by two people. Because this is uncomfortable, one of them would hop to another seat.
Figure 2.
(a) A gallium-doped p-type silicon semiconductor; (b) an arsenic-doped n-type silicon semiconductor.
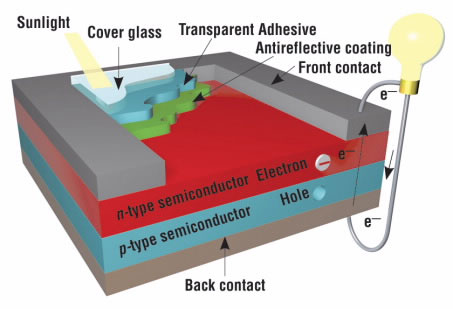
By placing a layer of p-doped silicon next to a layer of n-doped silicon, we create what is known in electronics as a p-n junction, or diode (Fig. 3). Diodes control electricity by only allowing current to flow in one direction. Imagine if our theater seats in the back with two people per seat and empty seats near the front. People would migrate from the back to the front—and not in the other direction. This is what happens in a p-n junction.
A solar cell is composed of many p-n junctions (fig. 3). When a solar cell is exposed to sunlight, the small particles that make up light, called photons, enter the solar cell and knock some of its electrons loose. When these electrons are in the border region — between the n-type and p-type layers — they move from the n-type to the p-type layer. Then, a metal wire collects these electrons and returns them to the back of the n-type layer through an external circuit, creating a flow of electricity.
Figure 3.
Schematic diagram of one layer of a solar cell, showing the n-type and p-type semiconductors.
On and off the grid
A major complication of solar energy is that it can be collected only when it is sunny. To solve this problem, most residential installations use a “net-metered” system. During the night or on cloudy days, a homeowner uses electricity from the electric grid — the network of power lines used to deliver electricity to houses and buildings — as usual. During a sunny day, the solar panels produce electricity that is used by the home, replacing electricity from the grid. If too much solar energy is collected by the solar panels — that is, if some of the solar energy is not used — it goes into the grid and earns the homeowner money, paid by the electric company.
Perhaps the most desirable way of using solar energy is for a home or building to be totally independent of the power grid. Rather than having a net-metered link to the power grid, the solar home or building would exist “off the grid.”
But what would happen during the night or on cloudy days? On sunny days, excess solar energy would be stored in large batteries and used in the absence of sunlight. An alternative is to use portable gas or diesel-powered generators to replace solar energy. With continued improvement in technology and lowered costs, chances are some day the United States will follow the model of the Utrik atoll and put solar power to work in a big way.
SELECTED REFERENCES
Solar 101: How solar works. Solarworld-USA:
http://www.solarworld-usa.com/solar-for-home/solar-101/history-of-solar.aspx. [Accessed April 2011].
Ewing, R. A.; Pratt, D. Got sun? Go solar: Get Free Renewable energy to Power Your Grid-Tied home, PixyJack Press, Masonville, Co.
Walsh, b. Solar power’s new style. Time. June 12, 2008.
Michael Tinnesand is a science writer and education consultant who lives in Portland, Ore. His latest ChemMatters article, A Single Ignition: A Cautionary Tale, appeared in the April 2011 issue.
Also in this Issue (October 2011)

Students Build Solar Homes
Every other year, students compete to build the world’s best solar homes. Check out what students have achieved in 2009 and what they will showcase this year at the Solar Decathlon in Washington, D.C.
Demystifying Gross Stuff
What’s behind acne, smelly breath, and flatulence? Bacteria and chemistry.

Lithium-Ion Batteries: A Clean Source of Energy?
Lithium-ion batteries power computers, iPods, and hybrid cars. But where does lithium come from? Are these batteries safe for the environment?
Subscribe to receive full issue!

