See how detergent interacts with food coloring and milk to make the colors mooove!

1. Carefully pour some milk in a dish so it just covers the bottom.
2. Gently add one or two drops of red, blue, and yellow food coloring to the same spot in the center of the milk.

3. Dip a cotton swab in your detergent, then gently touch the center of the food coloring. (Do not stir!)

4. Push the cotton swab down in the same spot all the way to the bottom of the plate and hold it there.

5. Dip a new cotton swab in the detergent. Then touch different areas of food coloring along the edge of the plate to see if the color will move again.
What to expect

The first time you touch the food coloring with detergent, the color should zoom to the edge of the plate. When you hold the cotton swab down longer, the color should swirl up from the bottom of the plate. When you touch the food coloring at the edge of the plate, the color should move around, mix, and make cool designs.
What else could you try?
We used liquid dish detergent to make the colors move. But you could try some other soap or detergent, like shampoo, liquid hand soap, laundry detergent, or regular soap. You could compare the way the different soap or detergent makes the colors move.

We used regular whole milk but you could try skim milk or half & half. Maybe the type of milk will make the colors move differently.

What’s happening in there?
Why did the colors move in cool ways? To answer this question, we have to use chemistry. Think about the atoms and molecules that the milk and detergent are made of and why they would act in this crazy way when they are mixed…

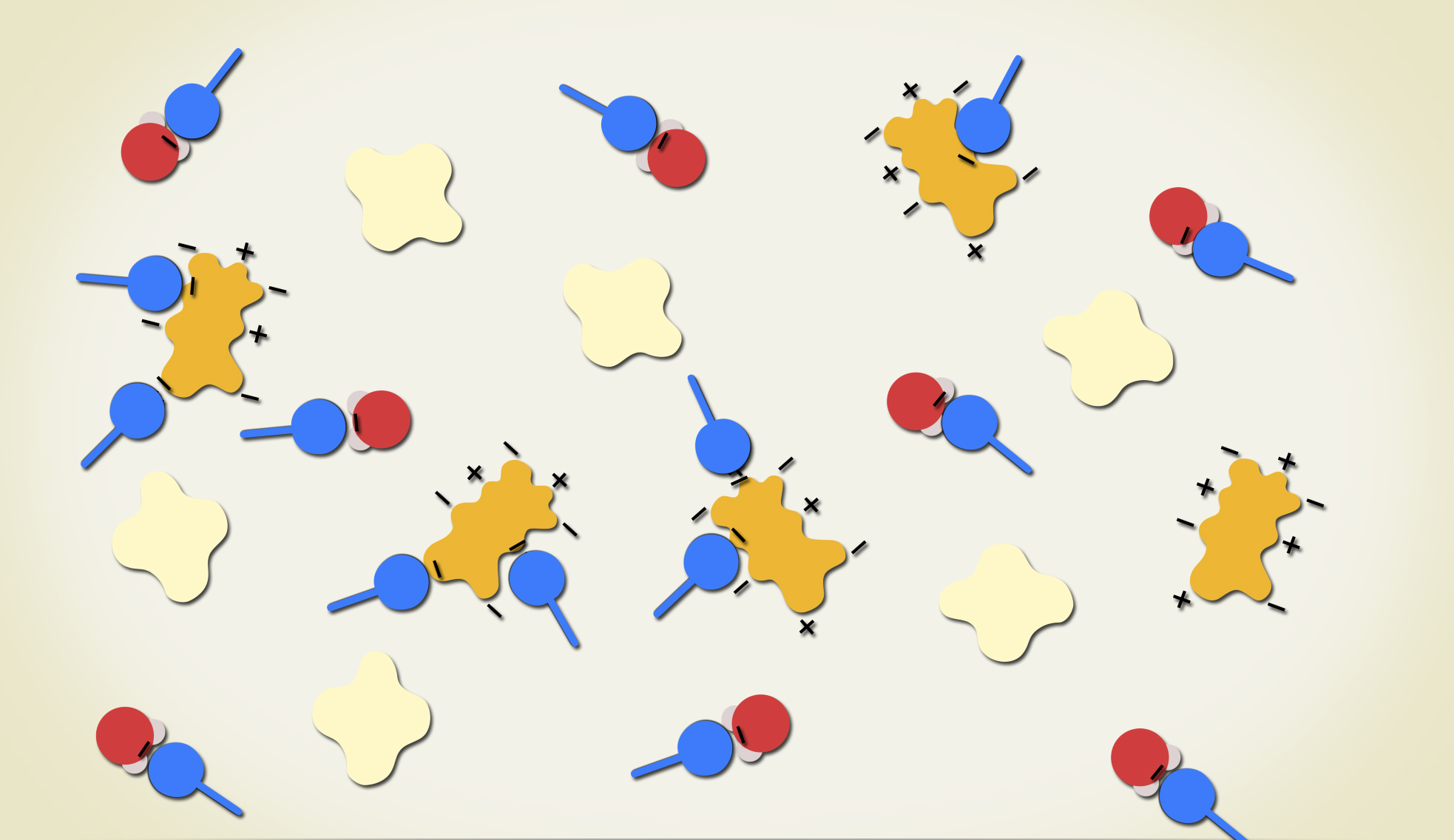
The milk is made mostly of water molecules, but it also has fat molecules and protein molecules which are bigger than water molecules.
The detergent is made up of detergent molecules.
The cool thing about the different molecules is that they have positive and negative charges in different areas. Some molecules or parts of molecules have no charges.
When you first put the detergent on the milk, the negative end of the detergent molecules line up with the positive end of the water molecules. This causes the detergent molecules to zoom out in every direction over the surface of the milk and push the food coloring out toward the edge of the plate.
And when you dip the detergent down into the milk or bring it to the food
coloring at the edge of the plate, there is usually some more swirling and movement of color.
The negative charge on the detergent molecules are attracted to the positive parts of the protein molecules. And the uncharged part of the detergent molecules lines up with the uncharged fat molecules.
There is a lot of moving as the detergent and other molecules twist and turn and switch around and different parts of the molecules come together. The food coloring molecules interact and ride along with all the other molecules, and that’s what makes the colors move!