EMBARGOED FOR RELEASE | April 10, 2013
Overcoming a major barrier to medical and other uses of ‘microrockets’ and ‘micromotors’
Note to journalists: Please report that this research was presented at a meeting of the American Chemical Society.
NEW ORLEANS, April 10, 2013 — An advance in micromotor technology akin to the invention of cars that fuel themselves from the pavement or air, rather than gasoline or batteries, is opening the door to broad new medical and industrial uses for these tiny devices, scientists said here today. Their update on development of the motors — so small that thousands would fit inside this “o” — was part of the 245th National Meeting & Exposition of the American Chemical Society, the world’s largest scientific society, being held here this week.
Joseph Wang, D.Sc., who leads research on the motors, said that efforts to build minute, self-powered robot devices have evoked memories of the 1966 science fiction film Fantastic Voyage. It featured a miniaturized submarine, which doctors injected into a patient. It then navigated through blood vessels to remove a blood clot in the brain.
Media Contact
During April 5-10, the contacts can be reached at 504-670-4707.
Michael Bernstein
202-872-6042
m_bernstein@acs.org
Michael Woods
202-872-6293
m_woods@acs.org
Fuel and propulsion systems have been a major barrier in moving science fiction closer to practical reality, Wang explained. Some micromotors and even-smaller nanomotors, for instance, have relied on hydrogen peroxide fuel, which could damage body cells. Others have needed complex magnetic or electronic gear to guide their movement.
“We have developed the first self-propelled micromotors and microrockets that use the surrounding natural environment as a source of fuel,” Wang said. “The stomach, for instance, has a strongly acid environment that helps digest food. Some of our microrockets use that acid as fuel, producing bubbles of hydrogen gas for thrust and propulsion. The use of biocompatible fuels is attractive for avoiding damage to healthy tissue in the body. We envision that these machines could someday perform microsurgery, clean clogged arteries or transport drugs to the right place in the body. But there are also possible uses in cleaning up oil spills, monitoring industrial processes and in national security.”
Wei Gao, a graduate student in Wang’s lab, described how the team at the University of California, San Diego, has developed two types of self-propelled vehicles — microrockets made of zinc and micromotors made of aluminum. The tubular zinc micromotor is one of the world’s fastest, able to move 100 times its 0.0004-inch length in just one second. That’s like a sprinter running 400 miles per hour. The zinc lining is biocompatible. It reacts with the hydrochloric acid in the stomach, which consists of hydrogen and chloride ions. It releases the hydrogen gas as a stream of tiny bubbles, which propel the motor forward. “This rocket would be ideal to deliver drugs or to capture diseased cells in the stomach,” said Gao.
Gao also described some of the latest advances in the technology. The newest vehicles are first-of-their-kind aluminum micromotors. One type, which also contains gallium, uses water as a fuel. It splits water to generate hydrogen bubbles, which move the motor. “About 70 percent of the human body is water, so this would be an ideal fuel for vehicles with medical uses, such as microsurgery,” said Wang. “They also could have uses in clinical diagnostic tests, in the environment and in security applications.”
Another type of aluminum micromotor doesn’t have gallium and is the first such motor that can use multiple fuels. “We’re really excited about this micromotor,” said Gao. “It is our most flexible one to date. For the first time, we’ve made a micromotor that can use three different fuels — acids, bases and hydrogen peroxide, depending upon its surroundings. Therefore, we can use these motors in many more environments than ever before.”
The scientists are working on extending the lifetimes of the vehicles so that they last longer and functionalizing them for specific biomedical applications. They also are exploring commercial partners for realizing real-life applications of this work, said Wang.
Wang acknowledged funding from the National Science Foundation and the U.S. Department of Energy.
To automatically receive news releases from the American Chemical Society contact newsroom@acs.org.
###
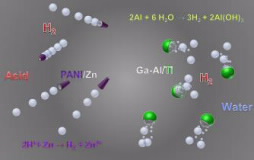
micromotors (right, green) could someday
deliver drugs, perform microsurgery or clean up
oil spills.

