FOR IMMEDIATE RELEASE
ACS News Service Weekly PressPac: January 10, 2007
ACS News Service Weekly PressPac: Jan. 10, 2007
Journalists’ Resources
Mark Your Calendars
Note to Journalists and Other Viewers
The American Chemical Society (ACS) News Service Weekly PressPac (PressPac) offers information on reports selected from 35 major peer-reviewed journals and Chemical & Engineering News.
This information is intended for your personal use in news gathering and reporting and should not be distributed to others. Anyone using advance ACS News Service Weekly PressPac information for stocks or securities dealing may be guilty of insider trading under the federal Securities Exchange Act of 1934.
Please cite the individual journal, or the American Chemical Society, as the source of this information.
News Items in this Edition
Related Links
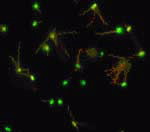
Poetry in motion may seem like an odd way to describe swimming bacteria, but that’s what researchers at Drexel University got when they enlisted Escherichia coli (E. coli) in an effort to tackle a major problem in developing lab-on-a-chip technology.
In a report scheduled for the Feb. 1 issue of ACS’ Analytical Chemistry, a semi-monthly journal, Min Jun Kim and Kenneth S. Breuer describe using E. coli to stir and enhance mixing in a controlled fashion in a microchannel. Blood and other medical samples may flow through such channels for analysis in future miniature laboratories etched on silicon chips. The researchers note, however, that getting those fluids to mix with chemicals in clinical tests and flow poses huge challenges because of difficulties in fabricating tiny pumps.
Journal Information
Journal: Analytical Chemistry
Journal Article: “Controlled Mixing in Microfluidic Systems Using Bacterial Chemotaxis”
Researchers thus are exploring biological motors, such as the spinning flagella that E. coli and other bacteria use to swim through fluids. In their experiments, Kim and Breuer harnessed the motion of E. coli flagella to achieve mixing in a fluid. The random motion of the micron-sized bacteria was used to enhance fluid mixing, and by adding chemical stimulants, the researchers were able to control both the direction and the magnitude of the stirring.
New scientific research may close a major loophole in the RNA world hypothesis, the idea that ribonucleic acid — not the fabled DNA that makes up genes in people and other animals — was the key to life’s emergence on Earth 4.6 billion years ago. That hypothesis states that RNA catalyzed all the biochemical reactions necessary to produce living organisms. Only later were those self-replicating RNA units joined by organisms based on DNA, which evolved into more advanced forms of life.
motility, power for
mixing
But how did ribonucleic acid appear? Scientists have shown that other organic compounds can form spontaneously under conditions believed to exist on the primordial Earth. The University of Manchester’s John D. Sutherland and colleagues point out, however, that no plausible prebiotic synthesis of ribonucleotides, the components of RNA, has been reported. His group offers the large part of such a potential synthesis in an article scheduled for the Jan. 17 issue of the Journal of the American Chemical Society, a weekly publication.
The researchers describe a process in which each of the two components for a ribonucleotide form in different locations on the primordial Earth. They combine when one evaporates and is delivered to the location of the second component in rainfall.
Journal Information
Journal: American Chemical Society
Journal Article: “Two-Step Potentially Prebiotic Synthesis of alpha-D-Cytidine-5’-phosphate from D-Gylceraldehyde-3-phosphate”
Why does the standard dose of certain medications prove dangerously high for some patients and too low to produce beneficial effects in others? Scientists have added a previously unrecognized factor to the list of explanations (such as age, gender, diet and genetics) for this common problem of individual variability in response to drugs.
Jeffrey P. Krise and Ryan S. Funk, at the University of Kansas, are reporting that variations in the body’s production of hydrogen peroxide — believed to serve as a signaling molecule at low levels — can affect accumulation of drugs inside cells. Their study, which involved cultured human cells and a common anti-cancer drug, is scheduled for the Feb. 5, 2007, issue of the ACS’s Molecular Pharmaceutics, a bi-monthly journal.
“The demonstrated correlation between hydrogen peroxide exposure and the concomitant increase in drug accumulation represents a substantial finding,” the study reports. “The results suggest that patients experiencing oxidative stress (or an increase in hydrogen peroxide levels) may have an increased response to a given dosage of a drug relative to a patient with decreased oxidative stress, or those patients chronically taking antioxidants [such as vitamins C or E] with their medications. This could be a very important factor in our continued efforts to provide more individualized dosing of drugs.”
Hydrogen peroxide’s effects could be especially important in about two dozen so-called narrow therapeutic index drugs (such as aminophylline, carbamazepine, lithium carbonate, phenytoin, theophylline and warfarin) for which very small changes in dosage level could cause either subtherapeutic or toxic results, they note.
Journal Information
Journal: Molecular Pharmaceutics
Journal Article: “Exposure of Cells to Hydrogen Peroxide Can Increase the Intracellular Accumulation of Drugs”
Scientists in Georgia are reporting successful lab tests of new polymer microparticles that show promise as a long-sought way to deliver drugs directly into the cell structures responsible for inflammation. Those immune system structures, macrophages, devour and destroy foreign substances such as invading bacteria and cellular debris. However, they also release so-called reactive oxygen species that help cause arthritis, acute liver failure and other inflammatory diseases.
In a report scheduled for the Jan. 17 issue of ACS’ Bioconjugate Chemistry, a bi-monthly journal, Georgia Tech’s Niren Murthy and colleagues at Emory University School of Medicine describe successful cell culture experiments with microparticles encapsulating superoxide dismutase (SOD). That compound is getting wide attention as a potential treatment for inflammatory diseases because it scavenges reactive oxygen species. One roadblock to clinical use of SOD, the researchers note, is the lack of a delivery system for SOD.
The new polymer microparticles have several advantages over other potential delivery systems, the researchers state. The particles remain intact until reaching acid environments such as the phagosomes — literal death chambers — that form after macrophages engulf bacteria and other particles. Then the polymers breakdown, releasing their SOD directly at the site where inflammation begins.
Journal Information
Journal: Bioconjugate Chemistry
Journal Article: “Polyketal Microparticles: A New Delivery Vehicle for Superoxide Dismutase”
Human embryonic stem cells have garnered extensive attention as potential future treatments for Parkinson’s and Alzheimer’s diseases, spinal cord injury, stroke, heart disease, diabetes and other diseases. Stem cells also may be the key to developing new treatments for certain types of cancer, according to an article scheduled for the Jan. 15 issue of Chemical & Engineering News (C&EN), the ACS’ weekly news magazine.
Written by C&EN Associate Editor Sarah Everts, the article describes how cancer stem cells are fostering a revolution in cancer research, with the realization that stem cells are key players in breast, blood, colon and bone cancer. Evidence suggests that conventional treatments kill ordinary tumor cells but spare the cancer stem cell, which continues to produce additional malignant cells.
The article is part of a C&EN cover story package on stem cells. Another part by Everts describes critical advances by chemists and other scientists in this high-profile field. Researchers, for instance, are defining the environmental conditions that are best for deriving and growing stem cells, including small molecules that can make a normal cell more like a stem cell. A third segment, by C&EN Associate Editor Susan R. Morrissey, discusses how the U.S. Patent & Trademark Office is reexamining several key patents previously granted for embryonic stem cells. Features on C&EN Online chart the business of stem cells and describe how scientists are working to use these cells as replacements for animal models in toxicological testing and disease research.
Journalists’ Resources
- ACS Chemical Biology
Highlights from the American Chemical Society journal, ACS Chemical Biology, are now available on EurekAlert!, the online science news service for reporters. ACS Chemical Biology is a monthly journal exploring cellular function from both chemical and biological perspectives. In addition to research papers and reviews, the journal also publishes “Spotlight” — current research in chemical biology from other journals; “Profile” — experts in the field; and “Points of View” — comments from leading scientists. The journal’s Web site is updated weekly with new content, and features a WIKI and an “Ask the Expert” section.
Mark Your Calendars
Chemistry has an increasingly important role in research on cancer diagnosis, prevention and treatment.
To spotlight that role, the American Chemical Society (ACS) and the American Association for Cancer Research (AACR) will cosponsor a special conference entitled Chemistry in Cancer Research: A Vital Partnership on Feb. 4-7 in San Diego, Calif.
The program will feature presentations by prominent scientists on drug discovery, proteomics, the chemical biology of carcinogenesis, biomarkers and analytical chemistry, modeling and bioinformatics, and structural biology.
The American Chemical Society — the world’s largest scientific society — is a nonprofit organization chartered by the U.S. Congress and a global leader in providing access to chemistry-related research through its multiple databases, peer-reviewed journals and scientific conferences. Its main offices are in Washington, D.C., and Columbus, Ohio.
Journal Information
Journal: Chemical & Engineering News
Journal Article: “Taming Stem Cells: Researchers are defining the chemical preferences of stem cells and figuring out what factors keep them eternal and which destine them to a differentiated fate”

