Water is a huge part of the earth, covering about 70 percent of the planet’s surface. Water is an essential part of every living thing on earth, and exists in three main states of matter. Solid water (ice) is in polar ice caps, glaciers, and snow. Liquid water is in oceans, lakes, rivers, and underground. Water as a gas, also called water vapor, is present in the air and clouds.
Heat from the sun changes solid water to liquid water (a process called melting). Heat also changes liquid water to water vapor (evaporation). As it cools, water vapor becomes liquid again (condensation). As it cools even more, liquid water becomes ice (freezing). Water molecules evaporate, condense, freeze, and melt — over and over. Throughout all these cycles, they are still water molecules.
Water molecules travel around the planet. Over and over, they evaporate, condense, freeze, and melt in what is called the water cycle. We can imagine some of the travels the water molecules might have taken.
For example, take the water that was in a drinking fountain near one of the first Earth Day celebrations. Water flows in the fountain and most goes down the drain, where it flows through pipes to the sewage treatment plant. After treatment, it would be released to a stream or river. The river could carry it to the ocean, where it might spend years. Eventually it might evaporate in the heat of the sun.
As the water evaporates, it leaves all the salts and minerals in the ocean behind. Our water now is a gas, so it could travel great distances. If it cools down, it condenses in the form of rain or snow, so maybe our water falls as snow in some mountain range, where it might freeze and remain for months or years. When it melts, it would flow into streams or rivers and might make its way back into a glass of water for someone to enjoy 50 years later!
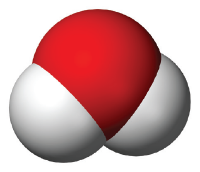
This is a model of a water molecule. The red represents one atom of the element oxygen, and the white represents two atoms of the element hydrogen. This is why you might hear someone call water “H2O”!
Water in the water cycle can have millions of different stories. Some water could be part of a polluted pond. In other places, it might be part of a glacier. Or the water could be inside a frog, a cloud, or a clump of soil. The water cycle helps keep us supplied with fresh, clean water. But sometimes the waste and pollution we produce contaminate our water sources — which is bad for all the plants and animals that depend on it.
And this is what Earth Day is all about. Water molecules were around in 1970 during the first Earth Day, and they have had an amazing journey ever since. Water is essential for all life on earth, and we all need to do what we can to keep it clean.
Susan Hershberger, Ph.D. is Director of the Center for Chemistry Education at Miami University in Oxford, Ohio.

