No-Hit Wonder! D3O®
By Brian Rohrig February 2017
A YouTube video titled “Smack Me On The Head With A Shovel!” shows a young man allowing himself to be bashed in the head repeatedly with a shovel. (Please do not try this at home!) The video is not promoting violence nor is it portraying some type of juvenile prank, but rather it is discussing some serious science. On the underside of the shovel is a flattened glob of bright orange goo that looks like Silly Putty. This amazing substance has the ability to absorb the energy of the moving shovel and is an exciting addition to the ultra-cool world of “smart materials.” Smart materials change in response to their environment. They provide the perfect example of how yesterday’s science fiction can become today’s reality.
This remarkable material goes by the trade name of D3O® and can be found in more than 100 products—everything from cell phone cases to kneepads to ballet shoes. While the name D3O® may sound like something out of Star Wars, its uses are much more down to earth. Its primary goal is to provide impact protection. D3O® was invented in 1999 by British engineer Richard Palmer. An avid snowboarder, he was looking for a way to protect himself from the many bumps and bruises he suffered from frequent wipeouts on his board. He found his answer in D3O,® which was named after the lab in which it was discovered. It is affectionately known by company insiders as “super goo” and comes in just one color—bright orange.
D3O® is an example of a unique class of materials known as non-Newtonian fluids. What makes these materials so different from regular fluids, and why are they used in so many everyday products?

Newtonian versus non-Newtonian fluids
Before discussing non-Newtonian fluids, let’s first examine what is meant by a “Newtonian” fluid. In addition to his many other discoveries, Isaac Newton did some groundbreaking work with fluids. He discovered that the viscosity of most fluids is affected only by temperature. Viscosity is the quantity that describes a fluid’s resistance to flow. A fluid with high viscosity resists motion while a fluid with low viscosity flows easily. More viscous substances, such as syrup and honey, take longer to pour than less viscous substances, such as water. (Be careful not to confuse viscosity with density. While cream may be thicker than milk, it is actually less dense, as cream will float on top of milk. )
Newton observed that if a substance is heated, it will become less viscous, and if cooled more viscous. When attempting to start your car on a sub-zero morning, you may notice that the engine has a difficult time turning over, as the oil in the car is thick and sluggish. Once the engine heats up, the oil becomes less viscous and flows more easily.
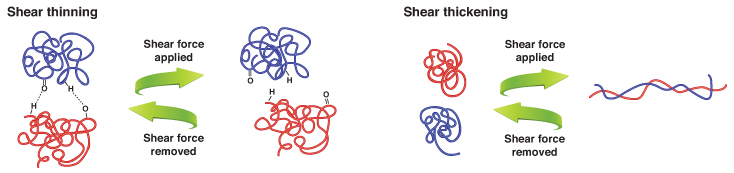
Most common fluids, such as water and oil, are Newtonian. Their viscosity remains constant no matter how fast they are forced to flow through a pipe or channel. So the only factor affecting their viscosity is temperature. But the viscosity of some fluids is affected by factors other than temperature. These fluids are termed non-Newtonian fluids. The viscosity of a non-Newtonian fluid will change due to agitation or pressure—technically known as shear stress. A shear stress will not affect the viscosity of a Newtonian fluid.
Non-Newtonian fluids are polymers. A polymer is composed of long chains of repeated units known as monomers that are strung together to yield giant macromolecules. The most common synthetic polymers are plastic, rubber, and fabrics, such as polyester and Spandex.TM Natural polymers include DNA, proteins, and starch.

Decreased viscosity
Consider ketchup. You may need to tap the bottom of the bottle or shake it loose to get it to come out. In doing so, you are applying a shear stress to the ketchup, causing it to become less viscous. Fluids that become less viscous when shaken or stirred are known as shear-thinning fluids. Other shear-thinning fluids are shaving cream, toothpaste and paint. If you rub shaving cream between your hands, it will become thin and runny, as its viscosity decreases.
The next time you brush your teeth, if you use an electric toothbrush, you will notice that as the bristles spin, agitating the toothpaste, you will begin to see the toothpaste twist and flow, becoming less viscous.
Another shear-thinning fluid, popularized in movies, is quicksand. If trapped in quicksand, the more you struggle, the faster you sink. Rather than helping you to escape, all of your thrashing about makes the quicksand less viscous, causing you to sink faster. But don’t worry—quicksand is seldom deep enough for you to sink in over your head. And since its density is about twice that of a person, if you relax you will float to the top.
Increased viscosity
Other types of fluids respond in the opposite way to a shear stress. If a shear stress is applied, they become more viscous. These fluids are termed shear-thickening. A mixture of corn starch and water is an excellent example of a shear-thickening fluid. When squeezed it appears to solidify. You can run across the surface of a swimming pool filled with a mixture of corn starch and water. When making gravy, which uses corn starch, it becomes thicker when you stir it.
Shear-thickening fluids can be found in your body. The synovial fluid that coats the joints in your knees and elbows is shear-thickening. Usually, this fluid is not very viscous, so as to allow for free movement of the joints. But if you bump your knee or elbow on a sharp corner of a table the synovial fluid comes to the rescue, instantly becoming more viscous and cushioning your joint from the painful blow.
D3O® is another type of shear-thickening fluid. It is composed of a polymer substance suspended in an oily-type liquid lubricant. This mixture is classified as a colloid, a mixture in which the dispersed substance is permanently suspended in a dispersing medium. Other common colloids include fog, whipped cream, and milk.
When a stress is applied slowly to a shear-thickening fluid, the polymer chains have time to move out of the way and rearrange themselves, thus the viscosity is not affected. But if a quick stress is applied, the polymer chains do not have time to rearrange and they become entangled, assuming a solid-like consistency as the viscosity greatly increases.
Imagine many cars trying to quickly leave through one exit in a parking lot. If everyone is in a hurry, the cars will become ensnarled in a traffic jam. But if the traffic exits slowly, there will be time for each car to leave in an orderly fashion.
Shear-thinning fluids behave in precisely the opposite way as shear-thickening fluids. Whereas a sudden stress will cause a shear-thickening fluid to harden instantly, a more prolonged force is required to thin a shear-thinning fluid.
Many applications of D3O®
D3O® can offer impact protection because it instantly becomes more viscous whenever a sudden shear stress is applied. One of its biggest uses is in protective knee and elbow pads.
In its normal state, D3O® behaves as a fluid, flowing easily. This free range of movement will increase the likelihood of the protective gear being worn, because it does not interfere with the user’s mobility. If you do take a tumble while skateboarding and your knee hits the ground, the bright orange D3O® putty will instantly harden, offering a greater degree of protection than traditional kneepads.

D3O® is also used to reduce concussions sustained by teen athletes. The American Academy of Pediatrics has reported a doubling of the number of reported concussions among teens in the past decade, with four to 5 million concussions occurring annually.
D3O® can be found in specially made helmets used in a wide variety of sports, from football to lacrosse to softball, with the hope that using this material can reduce concussion rates. Many helmets in use today primarily offer only impact protection, and do little to prevent the rotational acceleration that can lead to side-to-side or front-to-back movements of the brain within the skull, which can lead to concussions. Designing helmets that can reduce these types of movements is an active area of research, incorporating the fields of bioengineering, sports medicine, and materials science.

Perhaps the most important use of D3O® is to protect the lives of police officers and members of the military. Bulletproof vests and other types of body armor are often unpopular because they are too bulky and restrict movement, making it difficult to perform one’s job.
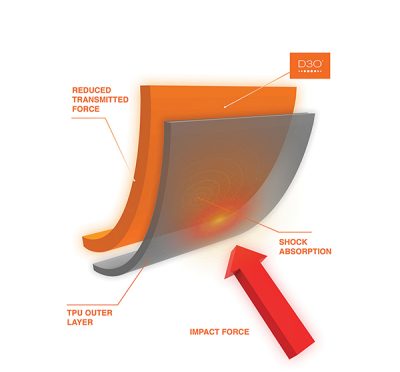
D3O® has been incorporated into bulletproof vests and helmets. It is more effective when layered with other substances, providing a very effective barrier that prevents penetration of projectiles such as bullets and shrapnel. If this type of body armor is impacted it instantly hardens, protecting the wearer.
If you drop your cell phone, a shattered screen or worse may be the end result. Incorporating D3O® in cell-phone cases provides excellent impact protection. These cases are easily identified by the bright orange stripes on their sides. If you tend to drop your phone a lot, you may want to consider this type of case.
The number of uses for D3O® and other similar non-Newtonian products is seemingly endless. They can protect you from serious injury and protect your most cherished possessions. A popular slogan embraced by the chemical industry is “Better Living through Chemistry.” D3O® provides a ringing endorsement of this motto.
Selected references
Woodford, C. Energy-Absorbing Plastics. Explain that Stuff, Oct 23, 2015:
http://www.explainthatstuff.com/energy-absorbing-materials.html [accessed Nov 2016].
Healy, M. Football Helmets and Concussion: A New Study Opens New Questions. Los Angeles Times, Feb 17, 2014:
http://www.latimes.com/science/sciencenow/la-sci-sn-football-helmets-concussion-20140217-story.html [accessed Nov 2016].
Zarda, B. The Incredibly Wide World of Smart Material D3O. Popular Science, Aug 14, 2009:
http://www.popsci.com/gear-amp-gadgets/article/2009-08/incredibly-wide-world-smart-material-d3o [accessed Nov 2016].
Brian Rohrig is a science writer who lives in Columbus, Ohio. His most recent ChemMatters article, “Piping Hot, Ice Cold… Thanks to Chemistry,” appeared in the December 2016/January 2017 issue.