Celebrating Paper: Why Paper Thrives in a Digital World
By Steve Keller April/May 2019
Teacher's Guides
Reading Supports | Tools and Resources

Even as e-textbooks and email replace some of their hard-copy counterparts, paper in its physical form is still everywhere. You can find it in nearly every room in the house, in your school, and where you work and play.
In fact, it might be hard to find a place where you won’t find paper, except maybe the forest where we get raw materials for paper. Even then, a paper map would come in handy if you wander outside your phone’s GPS signal range.
Although paper has become essential in everyday life, its properties are not well known. Yet they are what make paper the chameleon of materials. It can be clean and bright for us to write notes on, tough enough to form shipping boxes, and soft and absorbent enough to wipe our noses with.
Remarkably, for the vast array of useful characteristics paper exhibits, the composition of most paper is essentially the same: fibers derived from trees.
At the core of paper’s versatility are the fibers that have strength, flexibility, and the ability to bond to each other. Most papers that we encounter daily, such as printer paper, brown paperboard boxes, paper towels, or facial tissues, are assemblies of fibers that are almost entirely composed of sugars referred to as holocellulose. Holocellulose includes the long-chain polymer cellulose, and a variety of hemicelluloses that are shorter and may be branched.
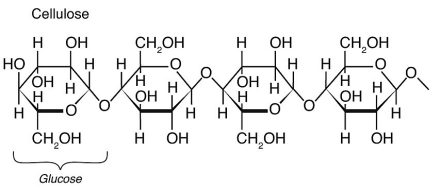
It’s the cellulose in particular that is responsible for the properties of paper that we value. Cellulose, the most abundant natural polymer, also gives distant cousins of paper—woven cotton textiles such as T-shirts and blue jeans—the durability, comfort, and water absorbency that we look for in clothing.
Strong, but not too strong
To understand how these natural fibers are used in paper, we need to take a look at their structure. Wood fibers generally come in two lengths. Longer fibers from softwoods, which come from evergreens, are 3 to 5 millimeters (mm) long. They are used to reinforce the paper, so it can withstand being pulled apart or torn. Hardwood fibers—which come from deciduous trees, the type that lose their leaves every year—are typically much shorter at about 1.2 mm in average length. They are used to fill in gaps and make paper smooth. Both hardwood and softwood fibers usually have widths less than that of a human hair, which measures about 50 micrometers across.
An even closer look shows us that the chemistry and microstructure of wood fibers are important to paper’s strength and flexibility. First, the long polymeric chains of cellulose, which have about 10,000 units of glucose (C6H12O6), hydrogen bond to each other and form strong, rigid crystalline regions. These structured components are connected to unstructured, flexible regions.
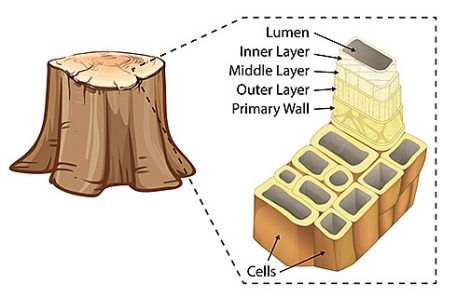
The long, skinny strands of connected cellulose form what are called microfibrils. Within trees and other plants, the microfibrils are found in the plants’ cell walls. The cell walls are composed of many layers of these microfibrils that are wrapped around an open center called the lumen (Fig. 1).
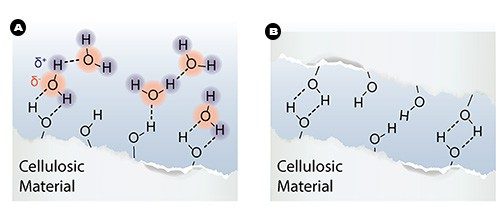
Due to its many polar hydroxyl (-OH) groups, cellulose is hydrophilic (“water-loving”). Water can be added and absorbed between the microfibrils, making the fiber wall softer and even more flexible (think of how dry spaghetti softens in boiling water) (Fig. 2a).
In papermaking, this flexibility allows the fibers to be flattened into ribbon-like structures as the paper is pressed. It can also easily conform to other fibers where they cross each other, further increasing the contact area between the fibers. This allows for hydrogen bonds to form between cellulose chains as the newly made paper dries (Fig. 2b).
For some paper products, such as toilet paper, hydrogen bonding between cellulose chains is all the strength that’s required for the final product. After use, these products should come apart easily. And since cellulose is hydrophilic, water can do the trick. It gets between the cellulose chains and causes the hydrogen bonds between cellulose molecules to break as new hydrogen bonds form between the cellulose and water. You can see this in action when toilet paper is immersed in water. Its structure breaks apart, and the fibers disperse.
This vulnerability to water comes in handy when we want to recycle other types of paper. Recycling facilities simply place recovered paper—for example, office papers, newspapers, or used corrugated containers—into a hydrapulper (a big blender) filled with water. The hydrapulper mixes the paper and water vigorously. The fibers easily separate from each other and can be cleaned and reused.

For sturdier stuff
Unlike toilet paper, products such as mail envelopes, shipping boxes, and office papers need to stand up to rain and the occasional splash of spilled water. At the same time, these products should be recyclable.
To add some resistance to the effects of water on hydrogen bonds between the cellulose chains in paper, the bonds need protection. This is where small amounts of substances called sizing agents come in. Sizing agents are organic compounds that are hydrophilic on one end and hydrophobic (“water-fearing”) on the other. When they’re added to the fibers, the hydrophilic ends covalently bond with cellulose during the drying process. Their hydrophobic ends face outward toward the surface of the paper and help slow down the absorption of water.
In some cases, paper manufacturers also apply a starch solution with micrometer-size hydrophobic particles to a paper surface. The starch forms hydrogen bonds with the cellulose, seals some of the pores, and coats some of the fiber-fiber bonds. The fine hydrophobic particulates slow the spreading of water on the fiber surfaces.
The Hydrogen Puzzle
Fillers and Fibers
Thanks to sizing agents, we have printer paper that won’t fall apart if a bit of rain or a glass of water spills on it. But in addition to varying degrees of durability, we’ve come to expect printer paper to be smooth and bright white—not the color of raw wood.
To achieve these texture and appearance requirements, manufacturers replace as much as 25% of the wood fiber with a filler composed of calcium carbonate (CaCO3) particles, which can be prepared synthetically or mined from the earth as marble or chalk.
The filler particles are very small, with a diameter of about 1/10th the width of a wood fiber. This small size increases the scattering of light to make paper appear whiter. Even with this extra load, paper maintains much of its strength and resistance to damage from water.

Although stronger than facial and toilet tissue, papers treated with sizing agents are still readily recycled under the right conditions. This commonly involves breaking the paper down into smaller pieces in a machine called a repulper. Additional equipment separates the remaining debris, such as staples or plastic. The fibers then pass through a refiner, which bends the fibers and reopens the cell walls. This allows them to swell and become more flexible. The fibers can then re-form bonds as they did when they were new.
Now that we’ve figured out how to make paper more water-resistant while maintaining recyclability, let’s look at a paper product designed to do the opposite. Paper towels need to be strong and absorb liquids quickly. To combine these traits into one product, manufacturers add chemicals called polymeric cross-linkers. These agents form covalent bonds between the cellulosic fibers, so that water can enter the structure without separating the fibers from each other. At the same time, the amount of bonding in paper towels is limited. This keeps the density low, so lots of internal spaces are available for liquids to flow into.
So, the next time you wipe up a spill with a paper towel, use a cardboard box, or take a test on paper, you can think about the chemistry that went into each of those useful products!
Steven Keller is a professor of chemical engineering at Miami University in Ohio.
Selected References
Paper: Science and Sustainability. International Paper Company: https://www.hammermill.com/paper-is-power-free-school-curriculum [accessed Feb 2019]
Smook, G. Handbook for Pulp and Paper Technologists. 4th Edition. TAPPI Press: Peachtree Corners, Ga., 2016.
Scott, W. Principles of Wet End Chemistry. TAPPI Press: Peachtree Corners, Ga. 1996.
Also in this Issue...
The Final Four
Scientists raced to complete the periodic table. Now what?
*Available in print issue only
Searching for Patterns of Life
Searching for Patterns of Life

Trees and Paper Are Made of Sugars?
Who doesn’t love a sugary treat: a slice of chocolate cake? Or a scoop of ice cream? But what are sugars? And why are they in our paper?
To answer that question, let’s start by looking at the larger category of molecules that sugar falls into: carbohydrates.
⋙ Carbohydrates are a category of biomolecules that includes sugars, starches, and fibers. They store energy, provide structure, and are found naturally in fruits, roots, grains, plant stems, and wood.
⋙ Monosaccharides, or simple sugars, are the simplest carbohydrates. They include glucose and fructose.
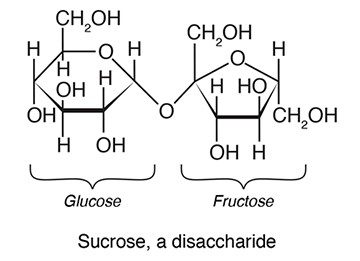
⋙ Disaccharides are made of two monosaccharides joined together. Sucrose, or table sugar, is one example. It consists of a glucose molecule joined to a fructose molecule.
⋙ Polysaccharides have three or more monosaccharides joined together. Cellulose, for example, consists of hundreds to thousands of repeating glucose units. It’s a major component of plants—and also an important part of dietary fiber!


