What molecules are we?
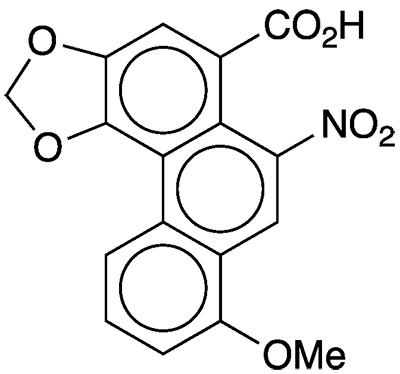
According to the Merck Index, aristolochic acids are “a group of 14 . . . substituted 1-phenanthrenecarboxylic acids . . . that occur in [the flowering plant family] Aristolochiaceae and in butterflies feeding on these plants.” Aristolochic acid I, shown here, is the most abundant of the group.
In 1953, H. Gänshirt at the Karlsruhe Institute of Technology (Germany) isolated aristolochic acids from several plants in the genus Aristolochia and determined their concentrations. In the following years, other researchers also isolated the acids, including M. Pailer and co-workers at the University of Vienna, who determined the chemical structures of several of the acids.
As shown in the hazard information table, aristolochic acids are highly toxic. They are suspected of causing the severe kidney disease Balkan endemic nephropathy (BEN) that is widespread in the Balkan Peninsula of Europe. A plant called birthwort (Aristolochia clematitis) is abundant in this region. It grows in wheatfields, where its contents enter the soil and groundwater and contaminate the crop. Epidemiologists estimate that ≈100,000 people are affected by BEN in the region.
But this is only a small part of the problem. As many as hundreds of millions or even billions of people may suffer from BEN worldwide. This is because Aristolochiaceae plants (genera Aristolochia and Asarum) have been used in traditional medicines for millennia, especially in Asia. In addition to BEN, individuals who consume these medicines can also develop aristolochic acid–induced liver cancer.
Aristolochic acid I hazard information
| Hazard class* | Hazard statement | |
|---|---|---|
| Acute toxicity, oral, category 3 | H301—Toxic if swallowed | |
| Acute toxicity, dermal, category 3 | H311—Toxic in contact with skin | |
Germ cell mutagenicity, category 1A | H340—May cause genetic defects | |
| Carcinogenicity, category 1A | H350—May cause cancer | |
*Globally Harmonized System of Classification and Labeling of Chemicals. Explanation of pictograms.
Molecule of the future
Rilpivirine is an HIC/AIDS drug developed by Tibotec (now part of Johnson & Johnson/Janssen Pharmaceuticals, Beerse, Belgium). It is a non-nucleoside reverse transcriptase inhibitor approved by the US Food and Drug Administration in 2008. It is a safer and more effective replacement for the older HIV medication efavirenz.
More recently, ViiV Healthcare1 (Brentford, UK) introduced another HIV drug, cabotegravir (shown), an HIV-1 integrase inhibitor.
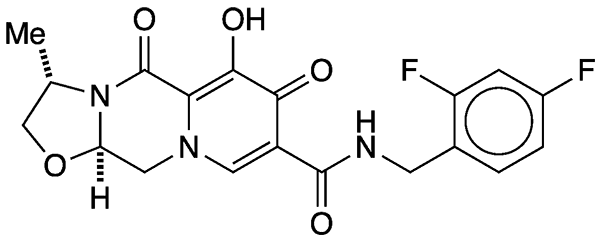
On January 21 of this year, FDA approved cabotegravir, alone and in combination with rilpivirine for treating HIV. The combination, called Cabenuva, is the first long-acting HIV treatment approved in the United States. The injectable formulation is given to patients once per month. Before they begin this regimen, patients are given cabotegravir and rilpivirine orally for 1 month to ensure that the drugs are well tolerated.
1. A joint venture 76.5% owned by GlaxoSmithKline.
Molecule of the Future
Once a month we bring you a newly discovered or developed molecule that has important implications for the future of chemistry or society in general. Look for it the third week of each month. Learn more about this month's Molecule of the Future below.
We're looking for more molecules of the future!
Do you have a suggestion for the next molecule of the future? Send your idea to MOTW.
Aristolochic acids
fast facts
| CAS Reg. No. | 313-67-7 |
| SciFinder nomenclature | Phenanthro[3,4-d]- 1,3-dioxole-5- carboxylic acid, 8- methoxy-6-nitro- |
| Empirical formula | C17H11NO7 |
| Molar mass | 341.27 g/mol |
| Appearance | Yellow crystals or powder |
| Melting point | 281–286 ºC (dec.) |
| Water solubility | ≈25 mg/L |
MOTW update
Ammonia was the Molecule of the Week for February 8, 2021. Its main use is in agriculture, but now it may have a “greener” use—as a transportation fuel. This past week, an article in Chemical & Engineering News presented an industrial update of the options and obstacles that will determine whether ammonia is indeed the “fuel of the future”.

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

