What molecule am I?
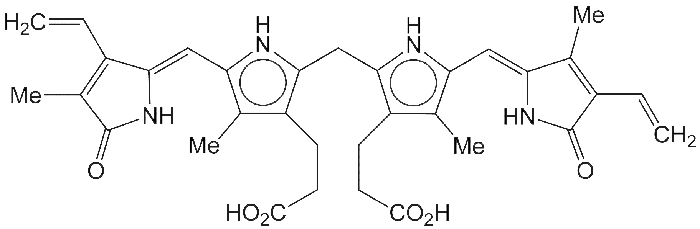
The bile pigment bilirubin is the major end product of the breakdown of heme in the liver. It is excreted in bile and urine. Its yellow color is responsible for the yellowish appearance of patients with jaundice.
In 1847, German scientist Rudolf Virchow and his colleagues isolated bilirubin crystals from hematomas and conjectured that it was derived from blood. More than a century later, Irving M. London and co-workers at Columbia University demonstrated that heme is indeed its source.
The central structure of heme is a porphyrin ring. Bilirubin’s structure is that of a ring-opened porphyrin: a chain of substituted pyrrole and 1,3-dihydro-2H-pyrrol-2-one rings.
The determination of bilirubin concentrations in blood is a key test for diagnosing medical conditions such as hepatobiliary or heart disease. Traditionally, blood tests have been performed in laboratories and can take several hours to complete. Patient-operated finger-stick tests with appropriate analytical equipment theoretically would take less time and be less expensive. But the recent Theranos debacle showed that this technology is not ready for prime time. Improvements in biosensors, sampling methods, and other factors are necessary to make this method practical.
MOTW Update
Glucose was the Molecule of the Week for July 12, 2012. Last week, C&EN reported that a handheld glucose meter can be used to determine whether a patient is suffering from the flu or a bacterial infection. If the patient has the flu, viral neuraminidases in a nasal swab liberate galactose in a test strip. Another enzyme decomposes the galactose to glucose, which registers on the meter. If no glucose is detected, it is presumed that the infection is bacterial.

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

