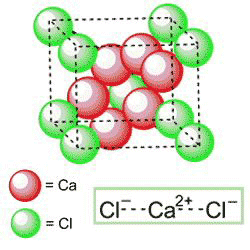
Calcium chloride (CaCl2) dissolves exothermally in water and is highly hygroscopic. These properties make it useful for applications such as removing water from gases and liquids, melting ice on roadways, maintaining a liquid layer on road surfaces for dust control, and preparing aqueous solutions with low freezing temperatures.

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

