What molecule am I?

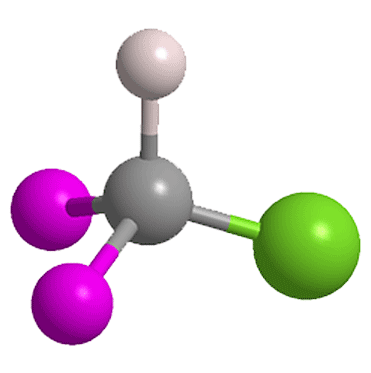
Chlorodifluoromethane (ClCF2H), also known as Freon 22, is a highly halogenated gas that can be used as a refrigerant or propellant. But it is a threat to the ozone layer, and under the Montreal Protocol it has been nearly phased out in the United States and other developed countries.
The phaseout restricted the utility of ClCF2H to the manufacture of fluorinated compounds (e.g., tetrafluoroethylene) until this year, when Xingang Zhang and colleagues at the Shanghai Institute of Organic Chemistry discovered that it can be used as a difluoromethylating agent. They found that, by using a transition-metal catalyst, the ClCF2 group can be coupled with aromatic boronic acids to form difluoromethyl-substituted aromatics.
In one example, the authors replaced the nitrile group in the herbicide cyhalofop-butyl with a ClCF2 group by converting the nitrile to a boronate ester and then treating the boronate with ClCF2H, the Pd2(dba)2* complex, and the Xantphos** ligand under relatively mild conditions (110 °C for 48 h). The yield of the difluoromethylation step was 90%, on the high end of the reactions given in this report.
The authors are currently attempting to discover the detailed mechanism of the difluoromethylation reaction and to reduce the amounts of reagents and catalyst required.
* dba = dibenzylideneacetone
** Xantphos = 4,5-bis(diphenylphosphino)-9,9-dimethylxanthene

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

