
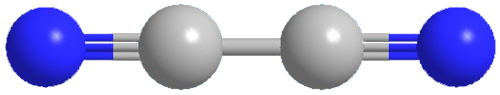
Cyanogen, the dinitrile of oxalic acid, is an extremely poisonous gas. It was first prepared as a pure substance by J. L. Gay-Lussac in 1815. It is usually made in the lab by oxidizing cyanide ion in aqueous solution; the industrial process is gas-phase oxidation of HCN. Cyanogen has few industrial uses, although NASA has considered using it as a rocket fuel.

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

