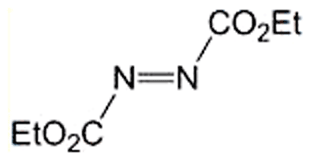
Our Halloween 2012 series begins with a molecule that has the acronym DEAD. It stands for diethyl azodicarboxylate, an orange–red liquid that becomes yellow when diluted in a solvent. DEAD is a strong electron acceptor; it oxidizes iodide to molecular iodine, hydrazine to molecular nitrogen, alcohols to aldehydes, and thiols to disulfides. DEAD’s principal use is as a reagent in the Mitsunobu condensation reaction, and it participates in Michael and Diels–Alder reactions. Because it explodes when heated, it cannot be shipped in pure form. Instead, it is transported in solution or adsorbed onto plastic particles.

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

