What molecule am I?
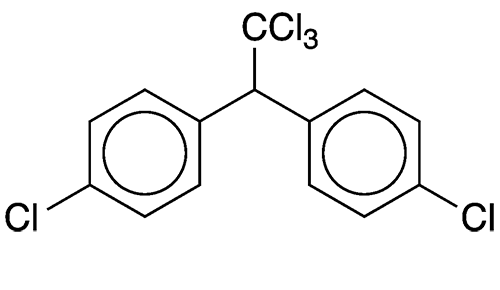
Dichlorodiphenyltrichloroethane (DDT) is a notorious insecticide that was considered to be a great scientific breakthrough. It goes by many other names, including the more formal 1,1’-(2,2,2-trichloroethylidene)bis(4-chlorobenzene) and 1,1,1-trichloro-2,2-bis(p-chlorophenyl)ethane.
DDT has been known since 1874, when Austrian chemist Othmar Zeidler synthesized it from chloral (trichloroacetaldehyde) and chlorobenzene. But it was not found to be a potent insecticide until 65 years later.
Swiss chemist Paul Hermann Müller, working at J. R. Geigy (Basel; now part of Novartis International), studied how insects absorb chemicals. Based on this research, he synthesized molecules that he thought would be taken up by insects and kill them. In 1939, after 4 years of research, the 350th compound Müller tested, DDT, killed a fly. For this breakthrough and subsequent work, he received the 1948 Nobel Prize in Physiology or Medicine.
DDT was an instant success against insects that carried disease and destroyed crops. It was used extensively during World War II and for three decades afterward to control insects that bore the pathogens for malaria and yellow fever. It is credited with saving the lives of millions of people who would likely have died from these diseases and others.
Soon after DDT was introduced, however, researchers found that it has a dark side. For humans and many other animal species, it acts as an acute and chronic toxin. It is an endocrine disruptor that can impair reproduction and harm the embryo or fetus. It is also a probable carcinogen. To make matters worse, it is not biodegradable and thus can build up in animal tissues with high lipid contents. Rachel Carson called attention to the dangers of DDT in her widely read 1962 book Silent Spring.
Carson’s work and subsequent findings led to the restricted use of DDT and, in 1972, an outright ban on its agricultural use in the United States. Under the Stockholm Convention on Persistent Organic Pollutants, it was banned worldwide in 2004; but it is still used, somewhat controversially, in emergencies against malaria outbreaks.
The ban on DDT is recognized as a major factor in preventing the extinction of the bald eagle and peregrine falcon in the United States. On January 10, Save the Eagles Day, you can celebrate the 2007 removal of the bald eagle from the endangered species list.
Dichlorodiphenyltrichloroethane hazard information
| Hazard class* | GHS code and hazard statement | |
|---|---|---|
| Acute toxicity, oral, category 3 | H301—Toxic if swallowed | |
| Acute toxicity, dermal , category 3 | H311—Toxic in contact with skin | |
| Carcinogenicity, category 2 | H351—Suspected of causing cancer | |
| Specific target organ toxicity, repeated exposure, oral, category 1 | H372—Causes damage to organs through prolonged or repeated exposure if swallowed | |
| Short-term (acute) aquatic hazard, category 1 | H400—Very toxic to aquatic life | |
| Long-term (chronic) aquatic hazard, category 1 | H410—Very toxic to aquatic life with long-lasting effects | |
*Globally Harmonized System (GHS) of Classification and Labeling of Chemicals. Explanation of pictograms.
This molecule was suggested by a reader. We present almost all of the molecules suggested by our readers. If you have a molecule you would like us to consider, please send us a message. And thank you for your interest in Molecule of the Week! —Ed.
Dichlorodiphenyltrichloroethane fast facts
| CAS Reg. No. | 50-29-3 |
| SciFinder nomenclature | Benzene, 1,1’-(2,2,2-trichloroethylidene)bis(4-chloro- |
| Empirical formula | C14H9Cl5 |
| Molar mass | 354.49 g/mol |
| Appearance | White crystals or powder |
| Melting point | 110.0–110.5 °C |
| Water solubility | 25 µg/L |
MOTW update:
March 11, 2024
Dichlorodiphenyltrichloroethane1 (DDT) was once the preeminent insecticide for agricultural and domestic use; but it was soon recognized as an acute and chronic toxin and a persistent environmental pollutant. It was banned in the United State in 1972 and worldwide in 2004.
But we haven’t seen the last of DDT. Last month, David L. Valentine and collaborators at the University of California, Santa Barbara, Oleolytics LLC (State College, PA), and the University of Southern California (Los Angeles) reported the results of a study of DDT contamination in deep San Pedro Basin sediments between the Port of Los Angeles and Catalina Island. Their findings indicated that
- bulk DDT waste disposal peaked in the 1950s;
- much of the waste is outside designated disposal areas;
- the waste includes substantial amounts of the DDT “daughter product” dichlorodiphenyldichloroethane (DDE)2;
- much of the waste was not disposed of in containers; and
- the company that dumped DDT also dumped low-level radioactive waste.
1. CAS Reg. No. 50-29-3.
2. CAS Reg. No. 72-54-8.

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

