What molecule am I?

March is MOTW Solvent Month! This is the last of four articles about key solvents—Ed.
Unlike the previous three solvents in this series, dihydrolevoglucosenone will not be familiar to many chemists. It is a recently developed, “green” alternative to existing dipolar aprotic solvents.
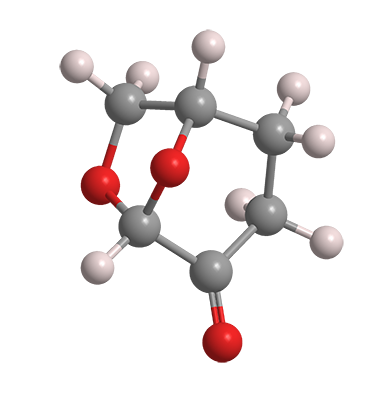
Formally named (1S,5R)-6,8-dioxabicyclo[3.2.1]octan-4-one, dihydrolevoglucosenone is derived from cellulose and is therefore chiral. It was developed by the Circa Group (Parkville, Australia), which is promoting it under the trade name Cyrene. In February, Circa, in a joint venture with Norske Skogindustrier ASA (Halden, Norway), opened a 50 t/year demonstration plant at Norske Skog’s papermill in southern Tasmania.
Dihydrolevoglucosenone is advertised as a biobased, biodegradable alternative to traditional polar aprotic solvents such as N,N-dimethylformamide and N-methyl-2-pyrrolidone, both of which pose toxicological and environmental hazards. Its low toxicity and high flash point (108 ºC) promise to make it a “safer” solvent if the price is right. In the past 5 years, several publications and patents illustrated the usefulness of dihydrolevoglucosenone as a solvent for the synthesis of organic compounds and polymers.
Dihydrolevoglucosenone hazard information
| GHS classification*: Serious eye irritation, category 2A | |
| H319—Causes serious eye irritation | |
*Globally Harmonized System of Classification and Labeling of Chemicals. Explanation of pictograms.
Dihydrolevoglucosenone
fast facts
| CAS Reg. No. | 53716-82-8 |
| Empirical formula | C6H8O3 |
| Molar mass | 128.13 g/mol |
| Appearance | Colorless to pale yellow liquid |
| Boiling point | 232 ºC |
| Water solubility | Miscible |
MOTW update:
October 4, 2021
Dihydrolevoglucosenone is a biobased, biodegradable solvent that has the potential to replace toxic aprotic solvents such as N,N-dimethylformamide and N-methyl-2-pyrrolidone. The product’s developer, Circa Group (Parkville, Australia), recently announced a site in northeastern France for a commercial plant that will make 1000 t/year of dihydrolevoglucosenone from waste cellulose.

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

