What molecule am I?
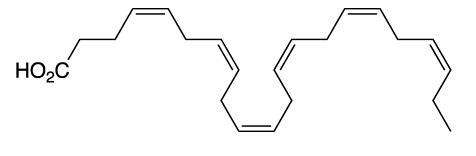
Docosahexaenoic acid (DHA) is a member of a group of natural compounds known to many as omega-3 fatty acids. The omega-3 (or ω-3) designation means that there is a double bond at the third carbon atom from the far end of the hydrocarbon chain. All six double bonds in DHA have the cis (Z) configuration, in contrast to fatty acids with trans double bonds.
DHA exists widely in the human body, particularly in the brain, skin, and retina. It is therefore not considered to be an “essential” fatty acid, such as α-linolenic acid (ALA) or linoleic acid, which the body cannot synthesize. Nevertheless, many dieticians recommend consuming external sources of DHA such as fish, algae, and dietary supplements. Supplementation is especially desirable for vegetarians and vegans.
The first recommended use of DHA as a supplement was for pregnant and lactating women. Recently, Wei Yang and collaborators at three medical research institutions in Beijing sought ways to overcome brain defects in newborn mice caused by Zika virus infections. They found that treating Zika-infected mice with DHA shortly after birth caused the mice to develop larger, heavier brains than control mice. The authors concluded that “our data might provide some potential therapeutic clues for [Zika] infections in pregnant women.”
Docosahexaenoic acid hazard information
| GHS* Hazard class | Hazard statement |
|---|---|
| Flammable liquids, category 4 | H227—Combustible liquid |
*Globally Harmonized System of Classification and Labeling of Chemicals.
Docosahexaenoic acid
fast facts
| CAS Reg. No. | 6217-54-5 |
| SciFinder nomenclature | 4,7,10,13,16,19-Docosahexaenoic acid, (4Z,7Z,10Z,13Z,16Z,19Z) |
| Empirical formula | C22H32O2 |
| Molar mass | 328.49 g/mol |
| Appearance | Light yellow oily liquid |
| Boiling point | 450–500 ºC (est.) |
| Water solubility | Insoluble |

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

