What molecule am I?

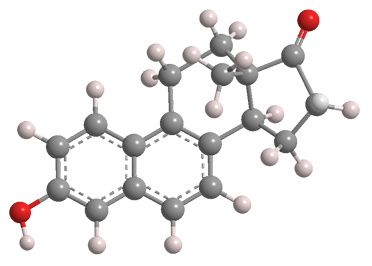
Equilenin is a steroidal estrogen originally isolated from the urine of pregnant mares. It was discovered by French researchers A. Girard and co-workers in 1932. The naturally occurring form is the (+)-isomer shown here. It should not be confused with equilin, in which the B ring of equilenin is monohydrogenated.
Among equilenin’s claims to fame is that it was the first complex natural product to be totally synthesized in the laboratory. The feat was accomplished in 1939 by W. E. Bachmann, Wayne Cole, and A. L. Wilds at the University of Michigan (Ann Arbor). The next year, the trio followed up with the total synthesis of the three non-natural stereoisomers of equilenin.
Equilenin, equilin, and several other conjugated equine estrogens (CEEs) are the components of Premarin*, a drug introduced by Wyeth (Philadelphia; now part of Pfizer) in 1941–42. Premarin is a hormone therapy primarily used to treat hot flashes, vaginal burning and dryness experienced by postmenopausal women who have undergone hysterectomies. But Premarin’s many adverse side effects caused it to be replaced by estradiol (Molecule of the Week for November 4, 2013) in some parts of the world.
CEEs are also used to treat some cancers; but ironically, equilenin and other CEEs are also suspected of causing cancer.
*From pregnant mare urine.
Equilenin hazard information
| GHS classification**: carcinogenicity, category 2 | |
| H351— Suspected of causing cancer | |
**Globally Harmonized System of Classification and Labeling of Chemicals. Explanation of pictograms.
Equilenin fast facts
| CAS Reg. No. | 517-09-9 |
| Molar mass | 266.33 g/mol |
| Empirical formula | C18H18O2 |
| Appearance | Pale beige crystals or powder |
| Melting point | 258–259 ºC |
| Water solubility | ≈1.5 mg/L |

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

