What molecule am I?
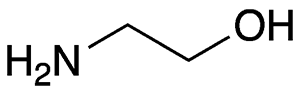
Ethanolamine, formally 2-aminoethanol, is a viscous, alkaline liquid with an unpleasant, ammonia-like odor. It is miscible in all proportions with water and several oxygenated organic solvents, including methanol, acetone, and glycerol. As the hazard information table shows, it is hazardous to humans and the environment.
In one of the earliest literature references to ethanolamine (1897), noted chemist Ludwig Knorr at the University of Jena (Germany) made the compound on a large scale by treating ethylene oxide with ammonia. The author cited work from as early as 1860 in which researchers made ethanolamine salts but could not isolate the free base. More recent ethanolamine research included synthesis from nitromethane and formaldehyde, but the prevailing manufacturing method still goes back to Knorr’s work.
Ethanolamine has several important industrial uses: as a “scrubber” to remove carbon dioxide, hydrogen sulfide, and other acidic pollutants from waste gas streams; as a starting material for manufacturing surfactants, chelating agents, and even pharmaceuticals; as an agent for softening leather; and as an additive for controlling pH in industrial water streams.
What’s new with ethanolamine? As with several recent Molecules of the Week, it has been discovered in outer space. Víctor M. Rivilla, Belén Tercero, Sergio Martín, and colleagues in Spain, Italy, Japan, Chile, and the United States identified ethanolamine in a molecular cloud in the interstellar medium, specifically in a complex in the Milky Way’s Galactic Center.
More than most molecules found in space, ethanolamine has particular relevance to possible origins of life on Earth. It is present in the water-soluble “heads” of phospholipids, which form all known cell membranes. And it could be a direct precursor of glycine, the simplest amino acid, which has been detected in the comet 67P/Churyumov–Gerasimenko.
Rivilla et al. conclude that their results “indicate that ethanolamine forms efficiently in space and, if delivered onto early Earth, could have contributed to the assembling and early evolution of primitive membranes.”
Ethanolamine hazard information
| Hazard class* | Hazard statement | |
|---|---|---|
| Flammable liquids, category 4 | H227—Combustible liquid | |
| Acute toxicity, oral, category 4 | H302—Harmful if swallowed | |
| Acute toxicity, dermal, category 4 | H312—Harmful in contact with skin | |
| Skin corrosion/irritation, category 1B | H314—Causes severe skin burns and eye damage | |
| Serious eye damage/eye irritation, category 1 | H318—Causes serious eye damage | |
| Acute toxicity, inhalation, category 4 | H332—Harmful if inhaled | |
| Specific target organ toxicity, single exposure, respiratory tract irritation, category 3 | H335—May cause respiratory irritation | |
| Short-term (acute) aquatic hazard, category 2 | H401—Toxic to aquatic life | |
| Long-term (chronic) aquatic hazard, category 3 | H412—Harmful to aquatic life with long-lasting effects | |
*Globally Harmonized System of Classification and Labeling of Chemicals.
Explanation of pictograms.
This molecule was suggested by a reader. We present almost all of the molecules suggested by our readers. If you have a molecule you would like us to consider, please send us a message. And thank you for your interest in Molecule of the Week! —Ed.
Ethanolamine fast facts
| CAS Reg. No. | 141-43-5 |
| SciFinder nomenclature | Ethanol, 2-amino- |
| Empirical formula | C2H7NO3 |
| Molar mass | 61.08 g/mol |
| Appearance | Viscous colorless liquid |
| Melting point | 10.5 °C |
| Boiling point | 171 °C |
| Water solubility | Miscible |

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

