What molecule am I?

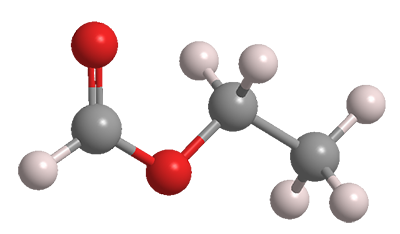
Ethyl formate is a simple aliphatic ester that is full of benefits and concerns. It’s a clear, slightly oily liquid with a pleasant, rumlike odor and the flavor of raspberries.
The ester occurs in many food substances, including fruits, coffee, tea, and grains. The synthetic product is often used as a flavoring agent in lemonade and artificial alcoholic beverages. It is used industrially as a solvent and as a fungicide and larvicide in processed foods such as dried fruits and cereals.
In its pure form, ethyl formate has several dangerous properties, as shown in the hazard information box (a new Molecule of the Week feature in 2018—Ed.).
In 2009, researchers at Cornell University and the University of Cologne detected ethyl formate in outer space—specifically, in the Sagittarius B2 dust cloud at the center of the Milky Way. Four years later, scientists in Spain and France confirmed its existence, this time in the Orion constellation. Discoveries of this and many other organic molecules in space give some researchers hope that extraterrestrial life can exist.
Ethyl formate hazard information
| GHS* classification: flammable liquids, category 2 | |
| H225—Highly flammable liquid and vapor | |
| GHS classification: acute toxicity, oral and inhalation, category 4 | |
| H302/H332—Harmful if swallowed or if inhaled | |
| GHS classification: eye irritation, category 2A | |
| H319—Causes serious eye irritation | |
| GHS classification: specific target organ toxicity—single exposure, category 3, respiratory system | |
| H335—May cause respiratory irritation | |
*Globally Harmonized System of Classification and Labeling of Chemicals. Explanation of pictograms.
Ethyl formate fast facts
| CAS Reg. No. | 109-94-4 |
| Molar mass | 74.08 g/mol |
| Formula | C3H6O2 |
| Appearance | Colorless liquid |
| Boiling point | 54 ºC |
| Water solubility | 88 g/L |

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

