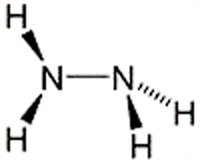
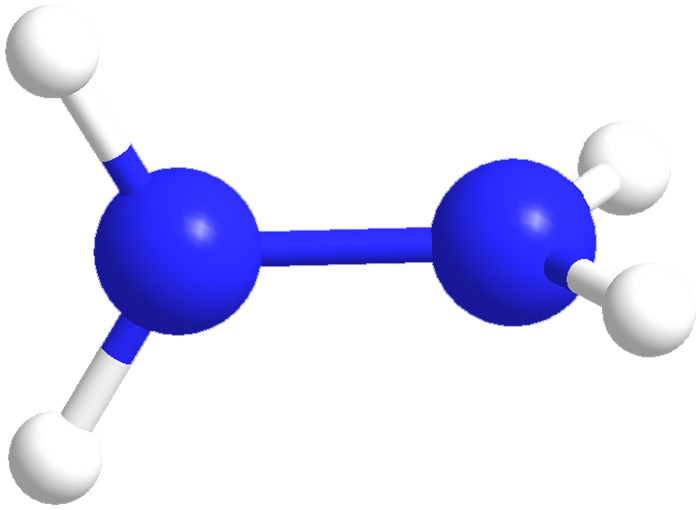
Hydrazine is an extremely toxic and unstable liquid with an ammonia-like odor. It has a wide explosive range (1.8–100%) and a low flash point of 52 °C. Its chief commercial use is as a blowing agent for plastics and metals; it also has many uses in synthesis and analytical procedures. Hydrazine has also been used as a rocket fuel, in part because it is hypergolic with N2O4, last week’s molecule. Recently, M. Stradiotto and R. J. Lundgren reported the use of a palladium catalyst to couple hydrazine with aryl groups.

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

