What molecule am I?

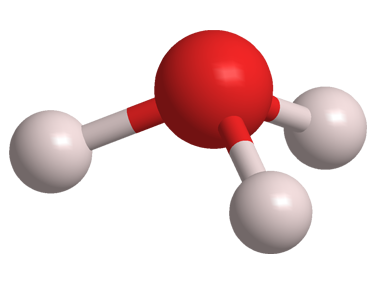
All acidic aqueous solutions contain protonated water, known commonly as the hydronium ion (H3O+). Brønsted acids release one or more of their protons (hydrogen ions), which combine with water molecules. Lewis acids extract one or more hydroxyl ions from water to release hydrogen ions, which, again, form hydronium ions.
So, when you see an aqueous chemical equation with the symbol H+, be assured that the actual ionic species is H3O+.
Why is “oxonium” the preferred name for the hydronium ion? It’s because hydronium is the simplest form of oxonium ions, in which three entities are attached to an oxygen atom, resulting in a net positive charge. [In my opinion, hydronium is much more descriptive than oxonium.—Ed.]
The concept of the hydronium ion has been known since the 19th century. In the 1880s, Swedish physicist/chemist Svante Arrhenius, working with German chemist Wilhelm Ostwald, defined an acid as a substance that dissociates in water to form hydrogen ions, which protonate water to form hydronium ions. Protonating acids became known as Arrhenius acids.
Arrhenius worked at Sweden’s Royal Institute of Technology (Stockholm) for his entire career. Later in his career, he helped establish the Nobel Prizes. He won the award in chemistry in 1903 for his definitions of acids and bases.
Not to be outdone, Ostwald won the 1909 Nobel Prize in Chemistry for his work in chemical reaction rates, equilibria, and catalysis. He worked at Riga Polytechnical Institute (Latvia) when he collaborated with Arrhenius.
Arrhenius’s ideas were later refined, independently, by Johannes Brønsted at the University of Copenhagen and Martin Lowry at the University of Cambridge (UK). Protonating acids are now known as Brønsted acids.
MOTW update
Phosphine was the Molecule of the Week for October 22, 2018. It is a toxic gas that is used in industrial synthesis. In recent years, phosphine was detected in outer space; some scientists suggest that it may be the source of phosphorus in biomoecules. Just in the past week, Jane S. Greaves at Cardiff University and the University of Cambridge (UK) and several colleagues worldwide reported the discovery of phosphine in the clouds that cover the planet Venus. This truly exciting finding was described in detail in major news outlets because of the possible presence of life on the shrouded planet.
This molecule was suggested by a reader. We present almost all of the molecules suggested by our readers. If you have a molecule you would like us to consider, please send us a message. And thank you for your interest in Molecule of the Week! —Ed.
Hydronium ion fast facts
| CAS Reg. No. | 13968-08-6 |
| SciFinder nomenclature | Oxonium |
| Empirical formula | H3O |
| Molar mass | 19.02 g/mol |
| Appearance | Cannot be isolated |
| Water solubility | Completely soluble |

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

