What molecule am I?
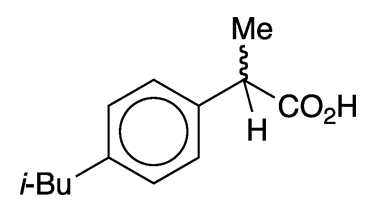
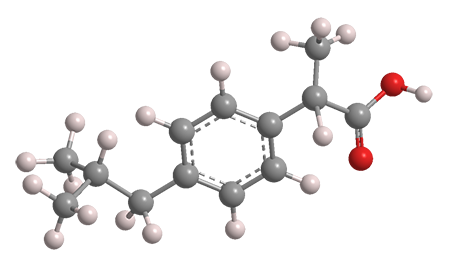
Ibuprofen is a frequently used over-the-counter drug for treating pain, inflammation, and fever. It is a relatively simple molecule that was discovered and developed in the 1950s and 1960s at Boots Pure Drug in Nottingham, UK (now Boots UK in Beeston). The initial British patent on ibuprofen and a long list of similar compounds was awarded to John Nicholson and Stewart Adams of Boots in 1964.
The trivial name ibuprofen comes from fragments of its chemical name, (±)-2-(p-isobutylphenyl)propionic acid. As the (±) implies, the article of commerce is racemic. The images show the more biologically active (S)-enantiomer. Ibuprofen is sold under a large number of trade names; Advil and Motrin are the most common in the United States.
In addition to its common uses, ibuprofen has been used to treat inflammatory diseases such as rheumatoid and juvenile idiopathic arthritis. But it is not without its risks: patients with cardiovascular disease, hypertension, and gastrointestinal disorders are cautioned against taking it. Ibuprofen also is contraindicated when individuals consume alcohol or low-dose aspirin.
Ibuprofen hazard information
| GHS classification*: acute toxicity, oral, category 4 | |
| H302—Harmful if swallowed | |
*Globally Harmonized System of Classification and Labeling of Chemicals. Explanation of pictograms.
Ibuprofen fast facts
| CAS Reg. No. | 15687-27-1 |
| Molar mass | 206.28 g/mol |
| Empirical formula | C13H18O2 |
| Appearance | Colorless crystals |
| Melting point | 75–77 ºC |
| Water solubility | 21 mg/L |
MOTW update
Clothianidin, imidacloprid, and thiamethoxam were the Molecules of the Week for February 17, 2014, August 25, 2014, and October 15, 2012, respectively. All three are neonicotinoid pesticides, and all three have been found to contribute to the worldwide die-off of honeybees. Use of these pesticides has been gradually restricted during the past several years, but now the European Union is about to ban them from all outdoor uses. Will the United States follow suit?

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

