What molecule am I?
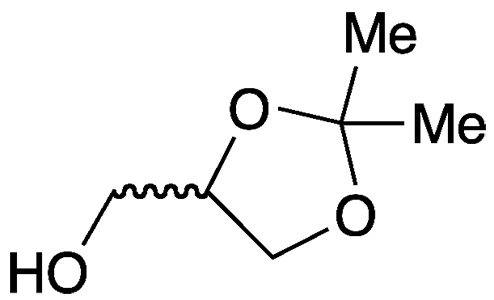
1,2-Isopropylideneglycerol (IPG) is a relatively high-boiling solvent and reagent that also goes by the names 2,2-dimethyl-1,3-dioxolane-4-methanol, acetone glycerol, and the trade name Solketal. It is miscible with water and organic solvents that range in polarity from alcohols to hydrocarbons. The molecule has a stereogenic center, but the article of commerce is normally the racemic mixture.
IPG was first synthesized in 1895 by legendary German organic chemist Emil Fischer1 during his research on the reactions of sugars with aldehydes and ketones. He and his colleagues treated anhydrous glycerol with acetone and a catalytic amount of hydrogen chloride. Researchers have continued to refine the synthesis; today, IPG is produced continuously using heterogeneous catalysis.
IPG is used as a starting material for producing glycerides by esterification of its hydroxyl group and transesterification of the isopropylidine moiety. It is also widely used as a solvent for chemical reactions and as a component of various types of cleaning solutions.
The wide availability of byproduct glycerol from biodiesel manufacture allows IPG producers to claim that it is a “green” solvent. As an example, the chemical giant Solvay makes it in Brazil under the trade name Augeo Clean Multi. In addition to the solvent’s sustainability, Solvay claims benefits such as low vapor pressure, slight odor, high flash point, and low human and environmental toxicity.
1. Fischer was the 1902 recipient of the Nobel Prize in Chemistry.
1,2-Isopropylideneglycerol hazard information
| Hazard class* | Hazard statement | |
|---|---|---|
| Flammable liquids, category 4 | H227—Combustible liquid | |
| Serious eye damage/eye irritation, category 2A | H319—Causes serious eye irritation | |
*Globally Harmonized System of Classification and Labeling of Chemicals.
Explanation of pictograms.

ALC-0315 (structure shown) is one of the ionizable lipids that is used to stabilize mRNA-based COVID-19 vaccines. It is produced by Acuitas Therapeutics (Vancouver, BC) and licensed to the Pfizer/BioNTech consortium. A similar lipid, SM-102, is used in the Moderna vaccine. Former Moderna executive Giuseppe Ciaramella calls them the unsung heroes of the success of the vaccines.
Systematic names: ALC-0315, decanoic acid, 2-hexyl-, 1,1′-{[(4-hydroxybutyl)imino]di-6,1-hexanediyl} ester; SM-102, octanoic acid, 8-{(2-hydroxyethyl)[6-oxo-6-(undecyloxy)hexyl]amino}-, 1-octylnonyl ester.
Molecule of the Future
Once a month we bring you a newly discovered or developed molecule that has important implications for the future of chemistry or society in general. Look for it the third week of each month. Learn more about this month's Molecule of the Future below.
We're looking for more molecules of the future!
Do you have a suggestion for the next molecule of the future? Send your idea to MOTW.
1,2-Isopropylideneglycerol
fast facts
| CAS Reg. No. | 100-79-8 |
| SciFinder nomenclature | 1,3-Dioxolane-4-methanol, 2,2-dimethyl- |
| Empirical formula | C6H12O3 |
| Molar mass | 132.16 g/mol |
| Appearance | Colorless oily liquid |
| Boiling point | 188–189 ºC |
| Water solubility | Miscible |

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

