What molecule am I?
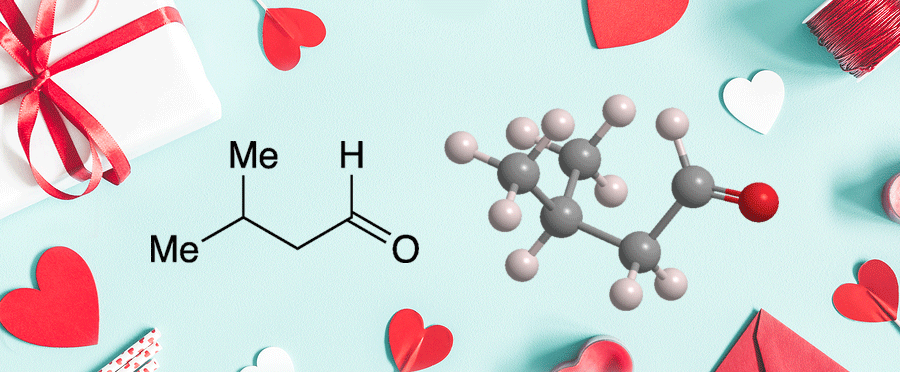
Isovaleraldehyde, formally 3-methylbutanal, is a pungent liquid that occurs in natural oils such as orange, lemon, peppermint, and eucalyptus. It is also found in processed foods such as beer, cheese, and chocolate. Its odor has been described variously from “apple-like” to “suffocating”.
In addition to its natural sources, isovaleraldehyde can be synthesized in several ways, including
- the oxidation of isoamyl alcohol with Na2Cr2O7 and H2SO4,
- the hydroformylation of isobutene,
- the isomerization of 3-methyl-3-butene-1-ol catalyzed by CuO–ZnO, and
- the reaction between isobutene and formaldehyde.
Isovaleraldehyde’s strong aroma makes it useful (in small amounts) as a perfume component. So . . . perfume, chocolate, and beer . . . what else could you possibly need for Valentine’s Day?
Isovaleraldehyde hazard information
| GHS classification*: flammable liquids, category 2 | |
| H225—Highly flammable liquid and vapor | |
| GHS classification: skin sensitization, category 1 | |
| H317—May cause an allergic skin reaction | |
| GHS classification: serious eye damage/eye irritation, category 2A | |
| H319—Causes serious eye irritation | |
| GHS classification: specific target organ toxicity, single exposure, respiratory tract irritation, category 3 | |
| H335—May cause respiratory irritation | |
| GHS classification: hazardous to the aquatic environment, long-term hazard, category 2 | |
| H411—Toxic to aquatic life with long-lasting effects | |
*Globally Harmonized System of Classification and Labeling of Chemicals. Explanation of pictograms.
Isovaleraldehyde fast facts
| CAS Reg. No. | 590-86-3 |
| Empirical formula | C5H10O |
| Molar mass | 86.13 g/mol |
| Appearance | Colorless liquid |
| Boiling point | 92–93 ºC |
| Water solubility | 1.4–20 g/La |
aConflicting data in literature

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

