What molecule am I?

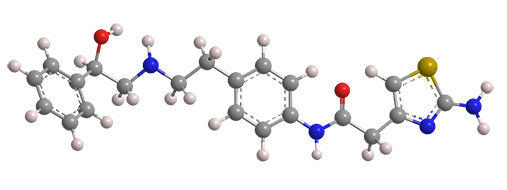
Mirabegron, a compound with multiple functional groups, is the active ingredient of drugs used for treating an overactive bladder. It is sold under the trade names Myrbetriq and Betmiga, both marketed by Astellas Pharma (Tokyo).
Mirabegron activates the β3 adrenergic receptor in the detrusor muscle of the bladder. Activating the receptor relaxes the muscle, allowing the bladder to accommodate a greater quantity of urine.
The drug, however, is not without adverse side effects. The most common is elevated blood pressure; but there are many others, including dry mouth, susceptibility to colds, and urinary tract infection.
Although mirabegron is taken orally, one of the hazards listed in its safety data sheets (see hazard information box) is oral toxicity. But the medicine is not toxic unless the patient consumes 5–10 times the normally prescribed dosage.
Mirabegron hazard information
| GHS classification*: acute toxicity, oral, category 4 | |
| H302—Harmful if swallowed | |
| GHS classification: skin sensitization, category 1 | |
| H317—May cause an allergic skin reaction | |
| GHS classification: serious eye irritation, category 2A | |
| H319—Causes serious eye irritation | |
| GHS classification: reproductive toxicity, category 2 | |
| H361—Suspected of damaging fertility or the unborn child | |
| GHS classification: specific target organ toxicity, repeated exposure, category 2 | |
| H373—Causes damage to organs through prolonged or repeated exposure | |
| GHS classification: hazardous to the aquatic environment, long-term hazard, category 1 | |
| H410—Very toxic to aquatic life with long-lasting effects | |
*Globally Harmonized System of Classification and Labeling of Chemicals. Explanation of pictograms.
Mirabegron fast facts
| CAS Reg. No. | 223673-61-8 |
| Molar mass | 396.51 g/mol |
| Empirical formula | C21H24N4O2S |
| Appearance | White to off-white crystals or powder |
| Melting point | 138–140 ºC |
| Water solubility | Insoluble |

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

