What molecule am I?
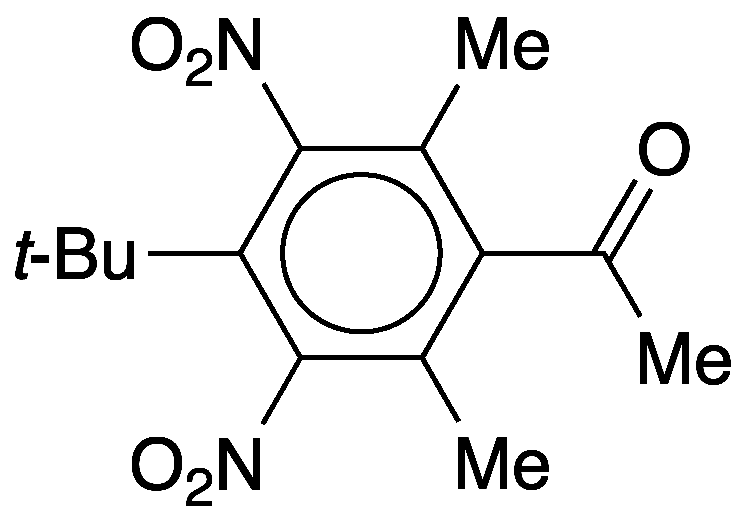
Musks are fragrant natural compounds produced by several animals and a few plant species. Animal gland–derived musks are so noticeable that many source animals have “musk” in their names: musk deer (Moschus spp.}, muskrat (Ondatra zibethicus), musk ox (Ovibos moschatus), and several others.
But musk fragrance is so highly desired in perfumes and cosmetics that natural sources are insufficient to meet the demand—not to mention the desire to avoid sacrificing animals for the sake of perfume. Chemists have met this challenge by synthesizing a series of artificial musks, including helvetolide, a monocyclic musk; galaxolide, a polycyclic musk; muscone, a macrocyclic musk; and musk ketone (4-tert-butyl-2,6-dimethyl-3,5-dinitroacetophenone), an aromatic musk that is this week’s featured molecule.
Musk ketone dates to 1888, when German chemist Albert Baur made it serendipitously while he was trying to find a better way to produce the explosive TNT. In the perfume industry, musk ketone is called a fixative because it stabilizes the volatility and improves the tenacity of perfume aromas.
Concerns about fragrance ingredients entering the environment have prompted regulatory agencies to require cosmetics manufacturers to disclose the constituents of their products. Until now, these formulations have been closely guarded trade secrets. This year, in advance of specific directives, the consumer products giant Unilever said it would voluntarily report such constituents, including musk ketone. The company said that ingredient lists will be available on its Web site sometime in 2018.
MOTW update:
December 5, 2022
Musk ketone1 was the Molecule of the Week for May 8. 2017. It is one of several natural and synthetic “musk” compounds used in the cosmetics industry. In November, Keiichi Yoshikawa and colleagues at Kao Corp. (Haga, Japan) and Duke University (Durham, NC) reported that four other “musk” molecules (musk xylene, mucsone, galaxolide, and helvetolide2), with diverse chemical structures, all activate the same olfactory receptor in humans. Their findings could help develop a unifying theory of musk odors.
1. CAS Reg. No. 81-14-1, 4.
2. CAS Reg. Nos. 81-15-2, 10403-00-6, 1222-05-5, and 141773-73-1, respectively.

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

