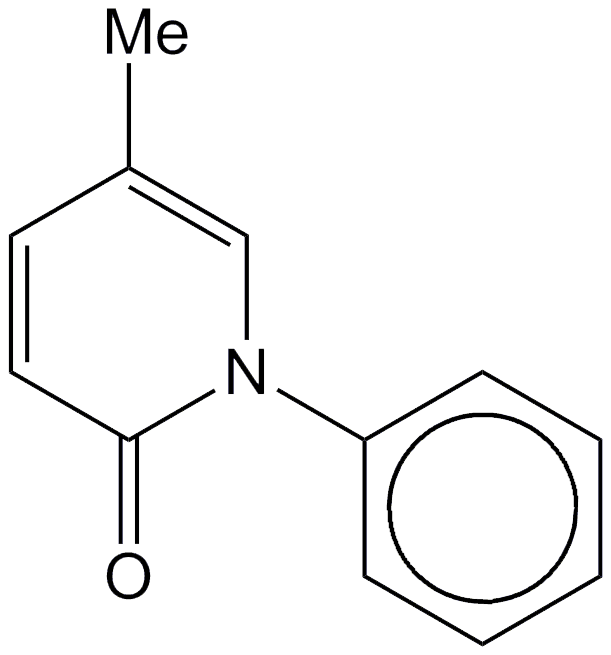
Idiopathic pulmonary fibrosis (IPF) is a fatal lung disease of unknown origin. Drug companies have sought a treatment for IPF for many years. Several concentrated on developing pirfenidone, including InterMune of Brisbane, CA; Shionogi of Osaka, Japan; and GNI Group of Tokyo. The drug was first approved for treatment in China in 2008, followed by approvals in India in 2010, Europe in 2011, Canada in 2012, and the United States and Mexico in 2014.
In August 2014, before pirfenidone was approved by the US Food and Drug Administration, Roche (Basel, Switzerland) paid US$8.3 billion to acquire InterMune. The product is expected to add US$1.6 billion to Roche’s annual sales by 2020.

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

