What molecule am I?
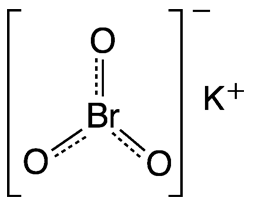
Potassium bromate (KBrO3) is a typical potassium salt in that it is water-soluble and has a high melting point. The bromate anion is a potent oxidizer.
KBrO3‘s only significant commercial use is as a “flour improver” in bakeries. According to the Professional Baker’s Reference in the King Arthur Flour Web site, it “strengthens dough and allows for greater oven spring and higher rising in the oven.” The reference states that KBrO3 is a “slow-acting” oxidizer, which means that it is effective during the mixing, fermentation, and proofing stages of the baking process.
Used properly, KBrO3 is completely consumed by the end of baking. But because good baking practices may not always be used, its carcinogenicity (see hazard information box), prompted its ban in the early 2000s in the European Union, Canada, China, South Korea, and some South American countries. Its use is still legal in the United States, but California’s Proposition 65 law dictates that bromated flour must be labeled as a carcinogen.
Potassium bromate hazard information
| GHS classification*: oxidizing solids, category 1 | |
| H271—May cause fire or explosion; strong oxidizer | |
| GHS classification: acute toxicity,oral, category 3 | |
| H301—Toxic if swallowed | |
| GHS classification: carcinogenicity, categories 1A, 1B | |
| H350—May cause cancer | |
*Globally Harmonized System of Classification and Labeling of Chemicals. Explanation of pictograms.
Potassium bromate
fast facts
| CAS Reg. No. | 7758-01-2 |
| Molar mass | 167.00 g/mol |
| Empirical formula | KBrO3 |
| Appearance | White crystals or powder |
| Melting point | 350 ºC |
| Water solubility | 75 g/L |

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

