What molecule am I?

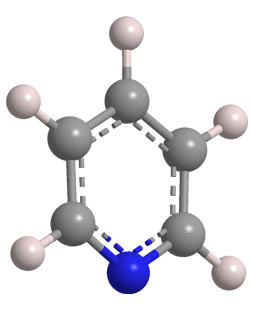
Pyridine is a colorless liquid with a foul odor and several hazardous properties. In the late 1840s, physician/chemist Thomas Anderson at the University of Edinburgh produced several liquids by heating animal bones to high temperatures. One of these turned out to be pyridine, which he named after the Greek word pyr (fire).
About 20 years later, chemists Wilhelm (aka Guglielmo) Körner at the University of Milan (Italy) and James Dewar at the University of Cambridge (UK), working separately, elucidated the structure of pyridine. The two chemists were friends, but they disagreed about which of them was the first to conceive of the structure. An interesting account of this episode is given by Alan J. Rocke at Case Western Reserve University (Cleveland) in a 1988 article.
In 1881, German chemist Arthur Rudolf Hantzsch at the University of Leipzig (Germany) developed a cumbersome, low-yielding synthesis of pyridine. Later, in 1924, Russian chemist Aleksei Chichibabin came up with an improved method, which is still in use today: a reaction between formaldehyde, acetaldehyde, and ammonia over a transition-metal fluoride catalyst to give dihydropyridine, followed by high-temperature catalytic oxidation to pyridine. Several other methods are also used, including the oxidative dealkylation of alkylpyridines.
Pyridine’s structure is isoelectronic with that of benzene, but its properties are quite different. Pyridine is completely miscible with water, whereas benzene is only slightly soluble. Like all hydrocarbons, benzene is neutral (in the acid–base sense), but because of its nitrogen atom, pyridine is a weak base.
In industry and in the lab, pyridine is used as a reaction solvent, particularly when its basicity is useful, and as a starting material for synthesizing some herbicides, fungicides, and antiseptics. Current worldwide pyridine production is ≈20,000 t/year, valued at about US$600 million.
Pyridine hazard information
| Hazard class* | Hazard statement | |
|---|---|---|
| Flammable liquids, category 2 | H225—Highly flammable liquid and vapor | |
| Acute toxicity, oral, category 4 | H302—Harmful if swallowed | |
| Acute toxicity, dermal, category 4 | H312—Harmful in contact with skin | |
| Skin corrosion/irritation, category 2 | H315—Causes skin irritation | |
| Serious eye damage/eye irritation, category 2A | H319—Causes serious eye irritation | |
| Acute toxicity, inhalation, category 4 | H332—Harmful if inhaled | |
| Carcinogenicity, category 2 | H351— Suspected of causing cancer | |
| Hazardous to the aquatic environment, acute hazard, category 3 | H402—Harmful to aquatic life | |
*Globally Harmonized System of Classification and Labeling of Chemicals.
Explanation of pictograms.
This molecule was suggested by a reader. We present almost all of the molecules suggested by our readers. If you have a molecule you would like us to consider, please send us a message. And thank you for your interest in Molecule of the Week! —Ed.
Pyridine fast facts
| CAS Reg. No. | 110-86-1 |
| SciFinder nomenclature | Pyridine |
| Empirical formula | C5H5N |
| Molar mass | 79.10 g/mol |
| Appearance | Colorless liquid |
| Boiling point | 115 ºC |
| Water solubility | Miscible |
MOTW update
Firefly luciferin was the Molecule of the Week for October 26, 2015. As its name implies, the molecule is essential for fireflies to emit light. The catalyst for the light-producing reaction is the enzyme luciferase, which may be useful in the quest for testing for COVID-19 infections and for developing drugs to combat the disease. Pei-Yong Shi and colleagues at the University of Texas Medical Branch at Galveston found that the bright light generated by luciferase allows them to visually detect antibodies against the SARS-CoV-2 virus several times faster than other methods. Likewise, the authors expect that luciferase will accelerate the development of therapies and vaccines against the virus.

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

