What molecules are we?

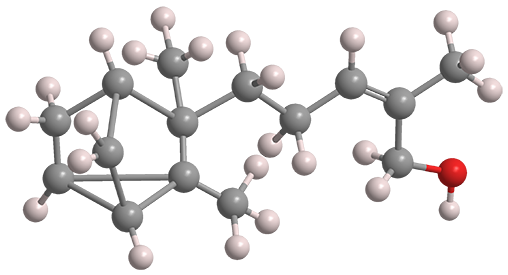

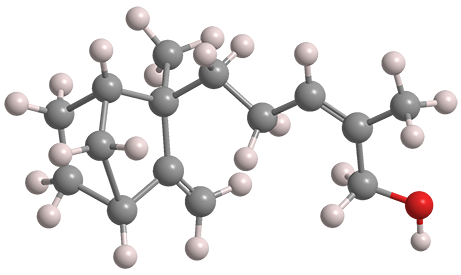
The santalol isomers are sesquiterpenes that comprise 70–90% of sandalwood oil, which is found in the heartwood of the tropical tree Santalum album and other members of that genus. The fragrant oil is valued as a component in perfumes.
Tricyclic α-santalol (above, left) is about three times as abundant as its bicyclic β-isomer (right). As the fast facts table shows, their properties are almost identical. Both have the Z-configuration at the side-chain double bond, although isomers with the E-configuration exist in small amounts in nature.
Friedrich W. Semmler at the University of Berlin conducted early research on isolating and identifying the santalols in the first decade of the 20th century. Shortly after, in 1914, V. Paolini and Laura Divizia at the University of Rome published an exceptionally detailed account of isolating the oil by steam-distilling sandalwood, separating the oil into α- and β-santalol and other components, and determining the properties of the santalols.
Initial attempts at the total synthesis of the santalols produced extremely low yields and/or imprecise stereochemistry of the products. It was not until the 1970s that organic chemists published highly stereoselective synthetic routes.
In 1970, future chemistry Nobelist Elias J. Corey and co-workers at Harvard University (Cambridge, MA) reported two syntheses of α-santalol: In one, they used a modified Wittig reaction to form the double bond in the side chain, starting from an available aldehyde derivative of the tricyclic structure. In the other, the researchers began with a tricyclic bromide and attached it to a lithiated propyne. After reduction of the triple bond to an olefine and subsequent functional group modification, α-santalol was recovered in good yield.
In 1979, Philip A. Christenson and Brian J. Willis* at Fritzsche Dodge and Olcott (New York City) reported a stereoselective synthesis of a mixture of β-santalol and its optical isomer. But not until 2009 did Charles Fehr and colleagues at Firmenich SA (Geneva, Switzerland) synthesize the natural (–)-enantiomer. The key step, as the authors put it, was a “highly selective copper‐catalyzed cyclization–fragmentation reaction of an enynol”.
Sandalwood oil is in high demand; but the natural product is very expensive, and chemical synthesis is not cost-effective. But Isobionics (Geleen, the Netherlands), a newly acquired subsidiary of BASF, has developed a biochemical process to make α- and β-santalol in about the same ratio as they occur naturally. The process uses the bacterium Rhodobacter sphaeroides to ferment cornstarch-derived sugars. In addition to saving costs (and sandalwood trees that would be destroyed otherwise), the company has made it possible to produce santalols wherever there is an inexpensive sugar source.
Santalol hazard information*
| Hazard class** | Hazard statement | |
|---|---|---|
| Skin sensitization, category 1B | H317—May cause an allergic skin reaction | |
*Mixture of α- and β-santalols.
**Globally Harmonized System of Classification and Labeling of Chemicals.
Explanation of pictograms.
α-Santalol fast facts
| CAS Reg. No. | 115-71-9 |
| SciFinder nomenclature | 2-Penten-1-ol, 5-[(1R,3R,6S)-2,3-dimethyltricyclo[2.2.1.02,6] hept-3-yl]-2-methyl-, (2Z)- |
| Empirical formula | C15H24O |
| Molar mass | 220.35 g/mol |
| Appearance | Pale yellow to yellow viscous liquid |
| Boiling point | 166–167 ºC (1.9 kPa) |
| Water solubility | Very slight |
β-Santalol fast facts
| CAS Reg. No. | 77-42-9 |
| SciFinder nomenclature | 2-Penten-1-ol, 2-methyl-5-[(1S,2R,4R)-2-methyl-3-methylenebicyclo[2.2.1] hept-2-yl]-, (2Z)- |
| Empirical formula | C15H24O |
| Molar mass | 220.35 g/mol |
| Appearance | Pale yellow to yellow viscous liquid or low-melting solid |
| Boiling point | 177.0–177.5 ºC (2.3 kPa) |
| Water solubility | Very slight |

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

