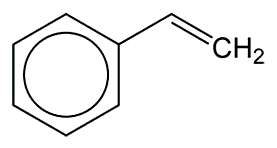
Styrene is a sweet-smelling, oily liquid that is almost insoluble in water but is miscible with most organic solvents. Its name comes from styrax (or storax) balsam, the resin of the Liquidambar genus of trees that grow in many places worldwide. M. Bonastre first isolated it in 1831 by distilling the balsam.
Styrene is among the most widely manufactured industrial chemicals: Production is approaching 30 million tonnes per year. Almost all of it is produced by dehydrogenating ethylbenzene; and almost all of it is used to make polystyrene or styrene copolymers, particularly styrene–butadiene rubber and latex. It polymerizes so readily that it forms a resin when it is exposed to the atmosphere.
Styrene has its downsides: It irritates the eyes and mucous membranes and causes more serious problems if it is inhaled or ingested. It has not been classified as a carcinogen, but its carcinogenicity is being evaluated by the US Environmental Protection Agency and the US Department of Health and Human Services. Styrene also contaminates water that flows through cured-in-place pipes.
Molecule of the Week Challenge:
In an alternative -styrene manufacturing process, what other chemical is coproduced with styrene? Send your answers to motw.@acs.org.
MOTW update:
April 02, 2018
Last week, C&EN reported that potentially harmful volatile organic compounds (VOCs) can be emitted from the plastics used in 3-D printers. Among the VOCs are former Molecules of the Week styrene (June 8, 2015), formaldehyde (May 29, 2006), and caprolactam (September 19, 2016). Regulatory standards may be in the offing for manufacturers of these printers.

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

