What molecule am I?
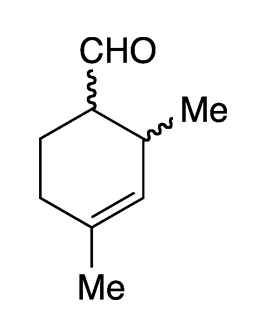
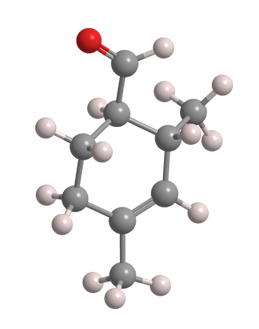
Trivertal (2,4-dimethyl-3-cyclohexenecarboxaldehyde) is an organic molecule with two chiral centers, as shown in the 2-D image. The manufactured product, a common fragrance ingredient, contains 95% of the two cis enantiomers and 5% of the trans isomers. In the fragrance industry, trivertal is called “2,4-ivy carbaldehyde”.
Commercial trivertal is likely contaminated with isomers that have the methyl groups in different positions on the ring. Thus, the melting point is depressed, and the mixture is a liquid. Purer forms are solids with a melting range of 85 to 90 ºC.
At the recent ACS National Meeting in San Diego, Guy Bertrand of the University of California, Riverside, spoke about his research group’s studies of stable cyclic (alkyl)(amino)carbenes, one of which was prepared from trivertal. The dimethylcyclohexene structure of trivertal and related compounds stabilizes the carbene, which can be used as a ligand for transition-metal catalysts.
As a proof of concept, the authors used the carbenes in gold(I) catalysts for the hydroamination of internal alkynes with secondary dialkylamines to synthesize complex quinoline derivatives in high yields. In his presentation, Bertrand discussed the mechanisms of the reactions that he reported earlier in a journal article.
Trivertal hazard information
| GHS classification*: flammable liquids, category 4 | |
| H227—Combustible liquid | |
| GHS classification: skin corrosion/irritation, category 2 | |
| H315—Causes skin irritation** | |
| GHS classification: skin sensitization, category 1 | |
| H317—May cause an allergic skin reaction** | |
| GHS classification: serious eye damage/eye irritation, category 2A | |
| H319—Causes serious eye irritation** | |
*Globally Harmonized System of Classification and Labeling of Chemicals. Explanation of pictograms.
**Not all material safety data sheets contain these warnings.
Trivertal fast facts
| CAS Reg. No. | 68039-49-6 |
| Empirical formula | C9H14O |
| Molar mass | 138.21 g/mol |
| Appearance | Colorless liquid |
| Boiling point | 196 ºC |
| Water solubility | ≈300 mg/L |

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

