EMBARGOED FOR RELEASE | March 21, 2010
Toward a three-in-one “dipstick” test for early detection of parasitic diseases
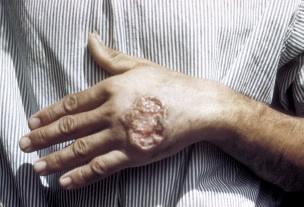
SAN FRANCISCO, March 21, 2010 — A new simple, inexpensive three-in-one test to diagnose a terrible trio of parasitic diseases that wreak havoc in the developing world is passing preliminary tests, scientists reported here today. Described during the 239th National Meeting of the American Chemical Society the test is for Chagas’ disease, leishmaniasis, and “sleeping sickness” or African trypanosomiasis.
This year will see about 800,000 new cases of Chagas disease, 2 million of leishmaniasis, and 70,000 of sleeping sickness (see sidebar). Most cases are discovered at a late stage, and together they cause tens of thousands of deaths each year and untold suffering. The drugs used to treat late-stage infections are often toxic and have potentially fatal side effects.
Media Contact:
Michael Bernstein
m_bernstein@acs.org
415-978-3504 (Meeting, March 21-25)
202-872-6042 (Before March 21)
Michael Woods
m_woods@acs.org
415-978-3504 (Meeting, March 21-25)
202-872-6293 (Before March 21)
“Early diagnosis is the key to improving treatment of these diseases,” said Ellen Beaulieu, Ph.D., a medicinal chemist in the Center for Infectious Diseases in the Biosciences Division of SRI International in Menlo Park, Calif., who reported on the test. “Diagnosis with conventional tests is difficult in developing countries where these diseases occur. We hope that our low-cost, simple test will play a role in helping poorer parts of the world combat these diseases and the poverty they engender.”
One existing test for diagnosing the three diseases involves taking a blood sample from a patient and examining it under a microscope for the parasites that cause the diseases. But the process is complex, requiring medical laboratory equipment and specially trained health care workers. Current blood tests are also time-consuming. In some cases, it can take up to 90 days to confirm results. Other tests detect the parasites using antibodies. But they require expensive electronic equipment that may not be available in remote areas of the developing world.
The new test exploits the common heritage of the parasites that cause Chagas, leishmaniasis, and African sleeping sickness. All three are closely related members of what scientists know as the trypanosomatidae family. Working together with Mary Tanga, Ph.D., Senior Director of Medicinal
Chemistry in SRI’s Biosciences Division, Beaulieu and colleagues developed special dyes that allow detection of an early disease stage, does not require the use of sophisticated lab equipment, and can produce results in as little as one hour.
Initial tests under laboratory conditions show that the dyes reveal the presence of the parasite marker and glow in ultraviolet light from a simple, handheld lamp. SRI researchers are making progress toward improving the sensitivity of the dyes.
The goal is to develop a “dipstick” version of the test that allows detection of the parasite metabolite using a simple paper strip like those used in urine tests for diabetes. Such a test could allow health workers in remote areas to diagnose the diseases by dipping the strip in a drop of blood and exposing it to ultraviolet light.
Silicon Valley-based SRI International is one of the world's leading independent research and technology development organizations. SRI’s Biosciences Division carries out basic research, drug discovery, and drug development, and provides contract services. SRI has all of the resources necessary to take R&D from Idea to IND®—from initial discovery to the start of human clinical trials—and specializes in cancer, immunology and inflammation, infectious disease, and neuroscience. SRI’s product pipeline has yielded marketed drugs, therapeutics currently in clinical trials, and additional programs in earlier stages. SRI is also working to create the next generation of technologies in areas such as diagnostics, drug delivery, medical devices, and systems biology.
###