What molecule am I?
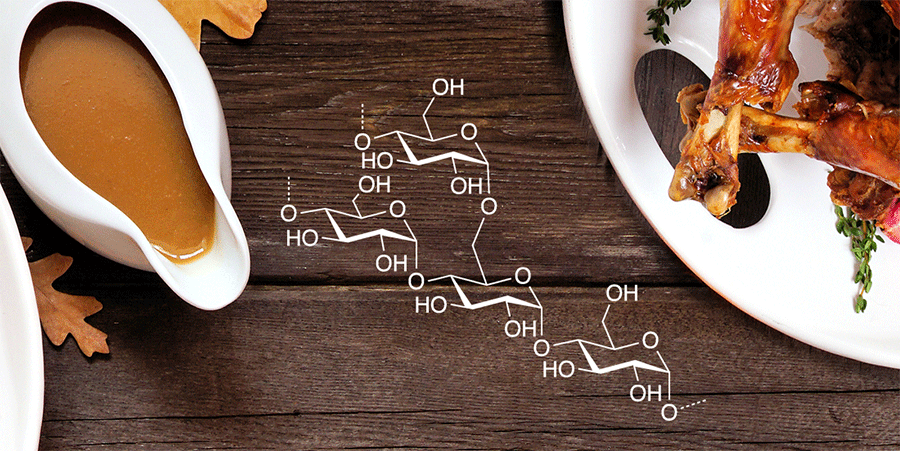
If you’re making gravy for your Thanksgiving meal, you’ll need some starch. Also, called amylum, starch is the component of plants that stores energy, much as animals store fats. Corn, wheat, and potatoes are especially rich in starch.
Starch consists of complex carbohydrates; the most abundant are amylopectin (≈70–80 wt% of starch) and amylose1 (the remaining ≈20–30 wt%). Both constituents are made up of chains of α-D-glucopyranosyl units. The difference between the two is that amylopectin is a branched-chain polymer that contains 1→4 and 1→6 α-D-glucopyranosyl linkages. Amylose is a helical polymer with only 1→4 α-D-glucopyranosyl linkages.
Amylopectin and amylose are insoluble in water; but as starch they do an excellent job of thickening and stabilizing water-based mixtures such as gravy, sauces, and soups. Amylopectin has excellent film-forming properties and is used to make edible food coatings. Its presence in starch also makes it useful for sizing textiles; it adds rigidity by reordering into crystalline structures. Science Direct has more information about the chemistry and uses of amylopectin.
When you’re preparing your favorite gravy recipe for your Thanksgiving stuffing, remember that it’s the amylopectin that makes it possible.
Amylopectin hazard information
| Hazard class* | GHS code and hazard statement |
|---|---|
| Not a hazardous substance or mixture |
*Globally Harmonized System (GHS) of Classification and Labeling of Chemicals.
Molecule of the Future
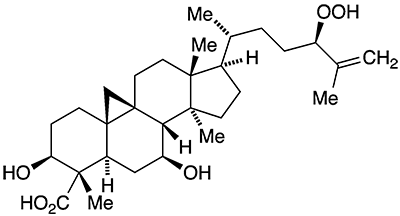
Castaneroxy A1 is a triterpenoid found in the leaves of the European chestnut tree Castanea sativa. In 2021, Cassandra L. Quave and colleagues at Emory University (Atlanta) and the University of Colorado Anschutz Medical Campus (Aurora) elucidated its structure and found that it has significant activity for reducing the virulence of methicillin-resistant Staphylococcus aureus (MRSA) skin infections. The authors also detected two other compounds in C. sativa leaves that have the same empirical formula as castaneroxy A.
1. CAS Reg. No. 2770688-76-9.
Molecule of the Future
Once a month we bring you a newly discovered or developed molecule that has important implications for the future of chemistry or society in general. Look for it the third week of each month. Learn more about this month's Molecule of the Future below.
We're looking for more molecules of the future!
Do you have a suggestion for the next molecule of the future? Send your idea to MOTW.
This molecule was suggested by a reader. We present almost all of the molecules suggested by our readers. If you have a molecule you would like us to consider, please send us a message. And thank you for your interest in Molecule of the Week! —Ed.
Amylopectin fast facts
| CAS Reg. No. | 9037-22-3 |
| SciFinder nomenclature | Amylopectin |
| Empirical formula | (C6H10O5)n |
| Molar mass | Variable; 107–108 g/mol |
| Appearance | White powder |
| Melting point | 160–166 °C |
| Water solubility | Insoluble |

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

