EMBARGOED FOR RELEASE | March 27, 2012
Blocking "oh-glick-nack” may improve long-term memory
Note to journalists: Please report that this research was presented at a meeting of the American Chemical Society
A press conference on this topic will be held at 7:30 p.m. Eastern Time, March 27, 2012, in the ACS Press Center, Room 15A, in the San Diego Convention Center. Reporters can attend in person or access live audio and video of the event and ask questions at www.ustream.tv/channel/acslive.
SAN DIEGO, March 27, 2012— Just as the familiar sugar in food can be bad for the teeth and waistline, another sugar has been implicated as a health menace and blocking its action may have benefits that include improving long-term memory in older people and treating cancer.
Media Contact
During the meeting, March 23-28, the contacts can be reached at 619-525-6268.
Michael Bernstein
202-872-6042
m_bernstein@acs.org
Michael Woods
202-872-6293
m_woods@acs.org
Progress toward finding such a blocker for the sugar — with the appropriately malicious-sounding name “oh-glick-nack” — was the topic of a report here today at the 243rd National Meeting & Exposition of the American Chemical Society (ACS), the world’s largest scientific society.
Linda Hsieh-Wilson, Ph.D., explained that the sugar is not table sugar (sucrose), but one of many other substances produced in the body’s cells that qualify as sugars from a chemical standpoint. Named O-linked beta-N-acetylglucosamine — or “O-GlcNAc” — it helps in orchestrating health and disease at their origins, inside the billions of cells that make up the body. O-GlcNAc does so by attaching to proteins that allow substances to pass in and out of the nucleus of cells, for instance, and helping decide whether certain genes are turned on or off. In doing so, O-GlcNAc sends signals that may be at the basis of cancer, diabetes, Alzheimer’s disease and other disorders. Research suggests, for instance, that proteins loaded up with too much O-GlcNAc can’t function normally.
At the ACS meeting, Hsieh-Wilson described how research in her lab at the California Institute of Technology and Howard Hughes Medical Institute implicate O-GlcNAc in memory loss and cancer. The research emerged from Hsieh-Wilson’s use of advanced lab tools for probing a body process that involves attachment of sugars like O-GlcNAc to proteins. Called protein glycosylation, it helps nerves and other cells communicate with each other in ways that keep the body coordinated and healthy. When O-GlcNAc is attached to a protein, that binding process is known as O-GlcNAc glycosylation.
Hsieh-Wilson’s team screened the entire mammalian brain for all O-GlcNAc-glycosylated proteins, using a new process that her laboratory developed. They identified more than 200 proteins bearing O-GlcNAc attachments or tags, many for the first time. The research was done in mice, stand-ins for humans in research that cannot be done on people. Some of the proteins carrying O-GlcNAc were involved in regulating processes like drug addiction and securing long-term storage of memories.
O-GlcNAc’s effects on one particular protein, CREB, got the scientists’ attention. CREB is a key substance that turns on and regulates the activity of genes. Many of the genes in cells are inactive at any given moment. Substances like CREB, termed transcription factors, turn genes on. Hsieh-Wilson found that when O-GlcNAc attached to CREB, CREB’s ability to turn on genes was impaired. When the researchers blocked O-GlcNAc from binding CREB, the mice developed long-term memories faster than normal mice.
Could blocking O-GlcNAc boost long-term memory in humans?
“We’re far from understanding what happens in humans,” Hsieh-Wilson emphasized. “Completely blocking O-GlcNAc might not be desirable. Do you really want to sustain all memories long-term, even of events that are best forgotten? How would blocking the sugar from binding to other proteins affect other body processes? There are a lot of unanswered questions. Nevertheless, this research could eventually lead to ways to improve memory.”
In a related study, Hsieh-Wilson found that O-GlcNAc interacted with another protein in ways that encourage the growth of cancer cells, suggesting that blocking its attachment might protect against cancer or slow the growth of cancer. And indeed, in mouse experiments, blocking O-GlcNAc resulted in much smaller tumors.
Again, a treatment for humans based on this discovery is far in the future, but the study singles out O-GlcNAc as a potential new target for developing anti-cancer drugs.
The scientists acknowledged funding from the National Institutes of Health (R01 GM084724-05 and 5T32 GM07616-30S1), Christopher & Dana Reeve Foundation and Howard Hughes Medical Institute.
To automatically receive news releases from the American Chemical Society contact newsroom@acs.org.
###
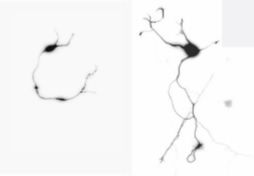
memory and even fight cancer. The neuron on
the left has CREB with O-GlcNAc and is short.
The neuron on the right does not have that
form of CREB and is long

