A Peek at Cometary Organic Chemistry
CHEMISTRY IN THE UNIVERSE
A Peek at Cometary Organic Chemistry
by Xin Su
December 14, 2015
When the Rosetta spacecraft was launched more than 10 years ago, it carried Philae, a robotic lander that contained a variety of instruments for measuring physical and chemical properties. On November 12, 2104, Philae touched down on the nucleus of comet 67P/Churyumov–Gerasimenko (see box), the first comet landing in the history of space exploration. On a mission to take a close look at the comet, Philae collected a great deal of information on its composition and properties.
Because many comets collided with Earth during its early stages, a popular theory for the origin of life is that primitive life forms, or at least prebiotic organic building blocks, reached Earth via collisions with comets and other small space objects. Therefore, a snapshot of the molecular composition of comets, particularly of organic substances, would be helpful for us to understand whether organic chemical reactions on comets tell us anything about the beginning of life.
Aldehydes in the atmosphere
One of the devices on Philae was Ptolemy, a gas chromatography–mass spectrometry instrument that was named after the Greek–Egyptian scientific pioneer Claudius Ptolemy. I. P. Wright and co-workers at the Open University (Milton Keynes, UK) reported and analyzed all six mass spectra collected by Ptolemy. The spectra contained much molecular information about the comet’s atmosphere: In addition to the expected signals of water, carbon dioxide, and carbon monoxide, the researchers identified CHO fragments, indicative of the presence of formaldehyde and acetaldehyde.
In addition, regular mass distributions seen in the mass spectra suggest the presence of repeating CH2O units. These results suggest the presence poly(oxymethylene) chains, which are likely the products of radiation-induced polymerization. In contrast, Ptolemy’s spectra did not reveal any aromatic compounds, such as benzene, or any sulfur-containing fragments. (Science DOI: 10.1126/science.aab0673)

| Comet 67P—Facts and Figures* | |
| Discovered by | Klim Ivanovych Churyumov Svetlana Ivanovna Gerasimenko |
| Discovery date | September 20, 1969 |
| Origin | Kuiper belt |
| Volume | 21.4 km3 |
| Mass | 1.0 ± 0.1 x 1013 kg |
*From Wikipedia.org
Nitrogen yes, Sulfur no
COSAC, short for Cometary Sampling and Composition, was another mass spectrometer onboard Philae. Unlike Ptolemy, COSAC consisted of a gas chromatograph and a time-of-flight mass spectrometer that could analyze materials supplied by the sample drilling and distribution system. A multinational team led by Fred Goesmann at the Max Planck Institute for Solar System Research (Göttingen, Germany) reported the results from COSAC.
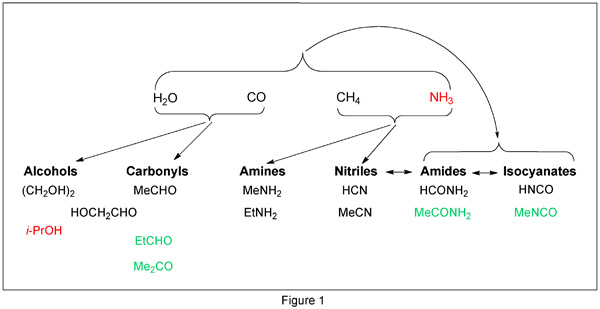
From the mass spectra, the authors assigned structures for 16 small molecules, all of which except for water and CO were organic compounds. Many nitrogen-containing compounds were identified, but the COSAC results confirmed the lack of sulfur-containing molecules. Figure 1 shows possible formation pathways for compounds identified by COSAC. The molecules shown in red were not “confidently” identified. Those in green—methyl isocyanate, acetone, propionaldehyde, and acetamide—had not previously been reported in comets. (Science DOI: 10.1126/science.aab0689)
Urea in outer space
These findings inspired chemists on Earth to take things a step further. A. H. H. Chang at National Dong Hwa University (Taiwan), R. I. Kaiser at the University of Hawaii (Honolulu), and their co-workers produced the biomolecule urea from ammonia–carbon monoxide ices in the presence of ionizing radiation. The ices were modeled after low-temperature interstellar and cometary ices such as those found on comet 67P.
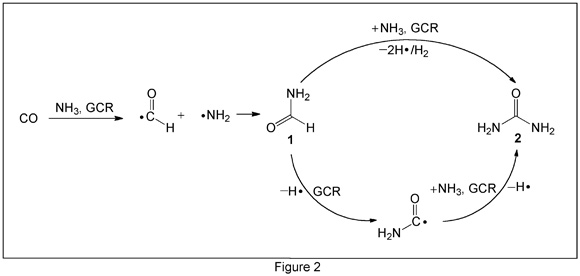
The researchers prepared a 500-nm-thick sheet of NH3 and CO in a 4:1 mol ratio and subjected it to 5 keV electron radiation, similar to the energy of galactic cosmic ray (GCR) particles. On-line and in situ infrared spectroscopy and mass spectrometry confirmed the abiotic formation of urea, along with formamide. From the COSAC data, formamide is the second most abundant gaseous compound on 67P, so it is reasonable to infer the existence of urea on the comet.
Figure 2 shows the authors’ proposed synthetic pathways from NH3 and CO to urea (2) via formamide (1). The upper pathway is concerted, whereas the lower one is stepwise. (Chem. Commun. DOI: 10.1039/c5cc07635h)
The big question
The discovery of rich, diverse organic chemistry on 67P is exciting, but it is still too early to establish any connection to the emergence of life forms. Sometime in the future, however, we will combine in situ molecular characterization in outer space with laboratory simulation experiments to find more answers to the ultimate question, “Where did we come from?”
Did You Notice?
Comet 67P/Churyumov–Gerasimenko was discovered exactly 2 months after the first moon landing!

