Lesson Overview for Teachers
View the video below to see what you and your students will do in this lesson.
Objective
Students will develop a model to describe that matter is made up of tiny particles, too small to be seen. Students will use the model to describe the differences in attraction among the particles of a solid, liquid, and gas. Finally, students will use their models of solids, liquids, and gases to explain their observations in the lesson.
Key Concepts
- Matter on Earth is in the form of solid, liquid, or gas.
- Solids, liquids, and gases are made of tiny particles called atoms and molecules.
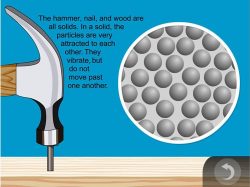
- In a solid, the particles are very attracted to each other. They are close together and vibrate in position but don’t move past one another.
- In a liquid, the particles are attracted to each other but not as much as they are in a solid. The particles of a liquid are close together, always moving, and can slide past one another.
- In a gas, the particles have very little attraction to each other. They are very far apart compared to the particles in a solid or liquid, and are constantly moving. The particles don’t interact with one another but just hit and bounce off of each other when they collide.
NGSS Alignment
- NGSS 5-PS1-1: Develop a model to describe that matter is made of particles too small to be seen.
Summary
- Students are introduced to the idea that matter is made up of tiny particles called atoms and molecules.
- Students observe a solid metal hammer and a nail and view a molecular model animation of the particles in a solid.
- Students squeeze a flexible plastic bottle with a balloon on top of the bottle to develop a model of the particles of a gas.
- Students also try to squeeze a bottle filled with water to develop a model of the particles of a liquid.
- Students watch a short animation that illustrates the incredibly tiny size of atoms and molecules.
- Finally, students make an argument that even though a mound of shaving cream keeps its shape, it is not a solid, and that even though sand takes the shape of its container, it is not a liquid.
Evaluation
Print the student activity sheet and distribute one per student when specified in the activity. The activity sheet will serve as the Evaluate component of the 5-E lesson plan.
Safety
Make sure you and your students wear properly fitting safety glasses or goggles.
Clean-up and Disposal
Remind students to wash their hands after completing the activities.
Save the bottles and sand for future use. All other common household or classroom materials can be saved or disposed of in the usual manner.
Materials
Materials for the Demonstration
- Hammer, nail, and wood
Materials for Each Group
- Flexible plastic soda bottle (18-20 oz.) with cap
- Balloon
Note: Flexible plastic soda bottles work better than disposable water bottles because water bottles are often not very flexible and tend to crush completely when squeezed.
Materials for the EXTRA EXTEND Demonstration
- Shaving cream
- Small plastic or paper plate
- Sand
- 2 clear plastic cups
Engage
1. Do a demonstration to show that a hammer is a hard solid.
Tell students that everything they can see and touch is called matter. Explain that all matter on Earth exists in the form of a solid, liquid, or gas, and that solids, liquids, and gases are all made of extremely tiny particles called atoms and molecules.
Tell students that an atom is the smallest building block of matter and a molecule is two or more atoms connected together. Atoms and molecules are so small that we can’t see them. Scientists use models to try to understand the behavior of atoms and molecules and to help explain the properties of matter.
Note: Even though atoms and molecules are different, for the purpose of this lesson, they both will be represented in the same way as a circle or sphere. In later lessons, they will be shown using different models.
Materials
- Hammer
- Nail
- Wood

Procedure
- Use a hammer to tap a nail partially into a piece of wood.
- Hold up the hammer and explain to students that solid, hard materials like the metal of the hammer are made of atoms that are very attracted to each other so they hold tightly together.
Note: If you don’t want to hammer a nail into wood, just tap the hammer lightly on a hard, sturdy, unbreakable surface, like a table or chair.
Show the animation Particles of a Solid.
Explain that the particles in a solid are very attracted to each other and vibrate in place. The strong attraction between particles keeps them close together and makes solids, such as the metal in the hammer, solid.
Give each student an Activity Sheet (PDF).
Students will record their observations and answer questions about the activity on the activity sheet.
Explore
2. Have students look at their “empty” bottle and then experiment with a balloon on it.
Question to investigate: Is an empty bottle really empty?
Materials for each group
- Flexible plastic soda bottle (18–20 oz.) with cap
- Balloon
Note: Plastic disposable water bottles don’t work as well because they are not very flexible and tend to crush completely when squeezed.

Procedure
- Have students look at and handle the uncapped bottle.
Ask students
- If the bottle has no liquid in it, is it completely empty?
Maybe. - Is there anything at all in the bottle?
Yes, there is air in the bottle.
Explain that the bottle has air in it and that air is made up of different gases like oxygen, nitrogen, and carbon dioxide that we breathe every day. Explain that a gas is made up of extremely tiny particles. Tell students that a gas is very different from a solid.
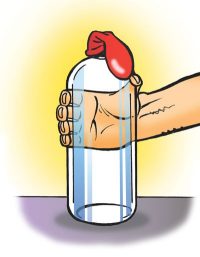
Tell students that if they still doubt that there is anything in the bottle, they can put a balloon on it and see what happens when they squeeze the bottle.
- Carefully put a balloon on the top of the bottle.
- Hold the bottle and squeeze it until you can’t squeeze it any further.
Ask students:
- What happens to the balloon when you squeeze the bottle?
The balloon expands.
- What do you think happened to the gas molecules when you squeezed the bottle and the balloon expanded?
The gas molecules must have been forced from the bottle into the balloon.
Explain
3. Show an animation and discuss how using the balloon shows that there must be something in the bottle.

Show the animation Particles of Gas in a Bottle.
Explain that the spheres represent the particles of a gas. Explain that the particles of a gas are not very attracted to one another and just hit each other and bounce off. The particles are also much farther apart than they are in a solid.
Explain that when the bottle is squeezed, the gas molecules move from the bottle into the balloon, making the balloon expand.
4. Have students squeeze the capped bottle.
Question to investigate: Can you force the molecules of a gas to move closer together?

Materials for each group
- Flexible plastic soda bottle (18–20 oz.) with cap
Procedure
- Take the balloon off of the bottle and put the cap on tightly.
- Squeeze the bottle.
Ask students:
- The bottle contains gas molecules. When the cap is on tight, were you able to squeeze the bottle?
Yes - You can’t squeeze a solid like metal or rock, so what is it about the molecules of a gas that allows you to squeeze it?
The molecules in a gas are very far apart so they can be squeezed together into a smaller space.

Show the animation Observing Gas in a Bottle.
Explain that with the lid on, the bottle can still be squeezed because gas molecules have a lot of space between them and can be compressed.
Explore
5. Have students investigate a bottle filled with water.
Question to investigate: Can you force the molecules of a liquid to move closer together?
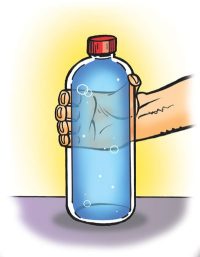
Materials for each group
- Flexible plastic soda bottle (18–20 oz.) with cap
- Water
Procedure
- Fill the bottle to the very top and place the cap securely on the bottle.
- Squeeze the bottle.
Expected results
The bottle is very hard to squeeze. It almost feels solid.
Ask students:
- Were you able to squeeze the bottle as much as when there was a gas in it?
No - After squeezing the bottle of gas and the bottle of liquid, would you say the particles of a liquid are closer together or farther apart than the particles of a gas?
Closer together
Explain
6. Show an animation and discuss the motion and arrangement of the particles of a liquid.
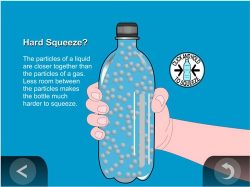
Show the animation Liquid in a Bottle.
Explain that the particles of a liquid are attracted much more than the particles of a gas and that they are much closer together. They are almost as close together as a solid but they can slide past each other. They are so close together that they are very difficult to squeeze.
7. Show an animation and compare the motion and arrangement of the particles of a solid, liquid, and gas.
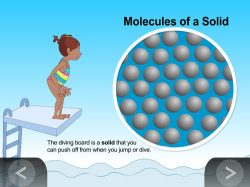
- Solid: Explain that in a solid, the particles are very attracted to each other so they are close together. The particles can move in their fixed positions but cannot slide past one another.
- Liquid: In a liquid, the particles are also attracted to each other but not as much as in a solid. The particles are close together, are constantly moving, and are able to slide past one another.
- Gas: In a gas, the particles are hardly attracted to each other. They are very far apart and just hit and bounce off each other as they constantly move about.
Extend
8. Show an animation and discuss how tiny atoms and molecules are.
Students have been introduced to the idea that matter, whether solid, liquid or gas, is made from tiny particles called atoms and molecules. Explain to students that the illustrations and animations of atoms and molecules they have seen are models used to represent atoms and molecules. Explain that the actual size of atoms and molecules is incredibly tiny, trillions of times smaller than the dots or spheres we use to represent them.
In fact, atoms and molecules are so small that millions of them would fit in the space of a single period at the end of a sentence.

Show the animation Atoms are Small—Really Small.
Tell students that the animation is about how incredibly small atoms and molecules are. It is based on the number of water molecules in a tablespoon of water, which is about 600,000,000,000,000,000,000,000 – About 600 billion trillion, so they are very small.
Extra Extend
9. Consider whether shaving cream and sand are solid, liquid, or gas.
Tell students that some books and other resources may define states of matter more simply in the following way:
- Solid – Maintains its shape even without a container
- Liquid – Takes the shape of its container
- Gas – Spreads out to fill any container
Question to investigate: How do you categorize a substance that doesn’t seem to fit these simple definitions for solids, liquids, and gases?

Materials for the demonstration
- Sand
- 2 clear plastic cups
- 1 can of shaving cream
- 1 small plastic or paper plate
Teacher Preparation:
- Put about ¼ to ½ cup of sand in one plastic cup.
- Put a small amount of shaving cream on a paper plate to use as a demonstration.
Is sand a liquid?
Procedure
- Carefully pour the sand from one cup to the other.
Ask students:
- Does the sand take the shape of the cup?
Yes. - Does that mean that sand is a liquid?
No. Each piece of sand is a solid but the pieces are so small that when you pour them, they take the shape of the container. Sand is a solid but acts sort of like a liquid because its tiny pieces take the shape of the container you pour it into.
Is shaving cream a solid?

Procedure, continued
1. Show students the mound of shaving cream.
Ask students:
- If you don’t touch it, does the mound of shaving cream maintain its shape without a container?
Yes - Does that mean it is a solid?
No. If you look very closely at the shaving cream, it is actually a liquid containing many tiny gas bubbles. So it is really a mixture of a liquid and a gas that together maintains its shape like a solid…until you touch it.





