
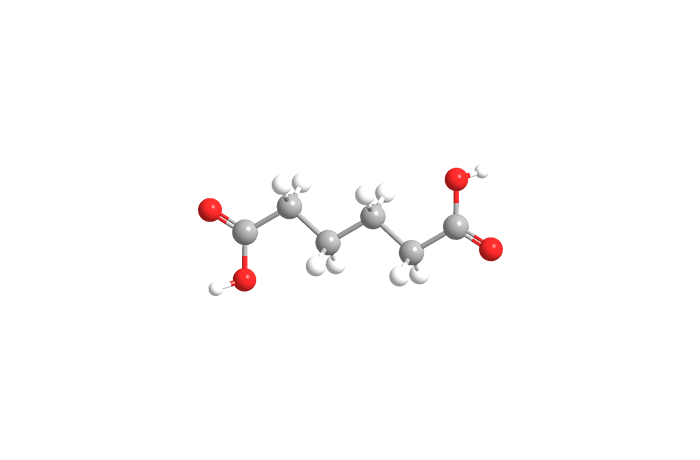
Adipic acid, or more formally hexanedioic acid, is a white crystalline solid that melts at 152 ºC. It is one of the most important monomers in the polymer industry.
Adipic acid is found in beet juice, but the article of commerce—≈2.5 million tonnes of it per year—is manufactured. In 1906, French chemists L. Bouveault and R. Locquin reported that adipic acid can be produced by oxidizing cyclohexanol. Today, the most common manufacturing process is the nitric acid (HNO3) oxidation of a cyclohexanol–cyclohexanone mixture called KA (for ketone–alcohol) oil.
Almost all adipic acid is used as a comonomer with hexamethylenediamine to produce nylon 6-6. It is also used to manufacture other polymers such as polyurethanes.
Using HNO3 to produce adipic acid has its downside: Copious amounts of nitrous oxide (N2O), a greenhouse gas, are coproduced and released into the atmosphere. In late 2014, K. C. Hwang and A. Sagadevan of National Tsing Hua University (Hsinchu City, Taiwan) reported a process that uses ozone and ultraviolet (UV) light to oxidize KA oil to adipic acid. This method eliminates the production of N2O; but before the process can be used commercially, problems associated with the formation of organic peroxides from ozone and the difficulty of using UV light on a large scale must be overcome.

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

