Thalidomide has a tragic history: It was introduced in Germany in 1957 as a sedative and hypnotic and was marketed over the counter largely as a drug for treating morning sickness in pregnant women. In the following few years, about 10,000 infants worldwide were born with phocomelia, or limb malformation. Only half of the infants survived, and some of those who did had other defects in addition to limb deficiencies. The thalidomide disaster caused many countries to tighten drug approval regulations.
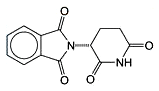
Thalidomide exists in two mirror-image forms: it is a racemic mixture of (R)- and (S)-enantiomers. The (R)-enantiomer, shown in the figure, has sedative effects, whereas the (S)-isomer is teratogenic. Under biological conditions, the isomers interconvert, so separating the isomers before use is ineffective.
More recently, thalidomide has proven useful for treating cancer and leprosy and is approved for these uses. But although more than 2000 papers have been written about its mechanism of teratogenic action, it was not until the past few years that this mechanism was established. In 2010, H. Handa and colleagues at the Tokyo Institute of Technology showed that its biological target is cereblon, a component of an E3 ubiquitin ligase complex. Earlier this year, N. H. Thomä and co-workers at the Friedrich Miescher Institute for Biomedical Research (Basel, Switzerland) determined the crystal structure of thalidomide bound to cereblon, which allowed them to characterize the mechanism.
Thalidomide was also the Molecule of the Week for May 17, 2010.
MOTW update:
March 11, 2024
Thalidomide1 is a drug introduced in the 1950s primarily for treating morning sickness in pregnant women. Tragically, it turned out to be a teratogen that caused about 10,000 infants worldwide to be born with phocomelia, or limb malformation. In later years, thalidomide was approved for treating cancer and leprosy.
In a recently published book, The Malta Thalidomide Affair, author Anatole Baldacchino, a prominent Maltese certified public accountant and member of several government boards of directors, writes about how slowly and poorly the Maltese government responded to the thalidomide crisis in that country. (Baldacchino has a personal stake in this story; he was born with relatively mild phocomelia.) The catastrophe came at a time when Malta was experiencing political, social, and religious upheaval. In January, The Dispatch, an American online magazine, published an article with a brief review of the book plus other troubling information about the thalidomide scandal.
1. CAS Reg. No. 50-35-1 (racemic).

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

