FOR IMMEDIATE RELEASE | August 20, 2017
Energized fabrics could keep soldiers warm and battle-ready in frigid climates
Note to journalists: Please report that this research will be presented at a meeting of the American Chemical Society.
A press conference on this topic will be held Tuesday, Aug. 22, at 10:30 a.m. Eastern time in the Walter E. Washington Convention Center. Reporters may check-in at the press center, Room 154A, or watch live on YouTube http://bit.ly/ACSLive_DC. To ask questions online, sign in with a Google account.
WASHINGTON, Aug. 20, 2017 — Soldiering in arctic conditions is tough. Protective clothing can be heavy and can cause overheating and sweating upon exertion. And hands and feet can grow numb despite wearing such gear. To keep military personnel more comfortable and battle-ready in bitterly cold climes, scientists are now conducting research aimed at creating high-tech fabrics that heat up when powered and that capture sweat. These fabrics could also conceivably make their way to consumer clothing in the future.
The researchers are presenting their results today at the 254th National Meeting & Exposition of the American Chemical Society (ACS). ACS, the world’s largest scientific society, is holding the meeting here through Thursday. It features nearly 9,400 presentations on a wide range of science topics.
Much of the Army’s cold-weather hand gear was designed more than 30 years ago, so soldiers often opt to buy winter gloves at retail stores, Paola D'Angelo, Ph.D., says. But even this modern gear isn’t enough to prevent paratroopers from losing feeling in their hands and feet when they parachute to earth in arctic conditions. “That’s problematic if soldiers have to operate weapons as soon as they land,” D’Angelo notes. “So we want to pursue this fundamental research to see if we can modify hand wear for that extreme cold weather.”
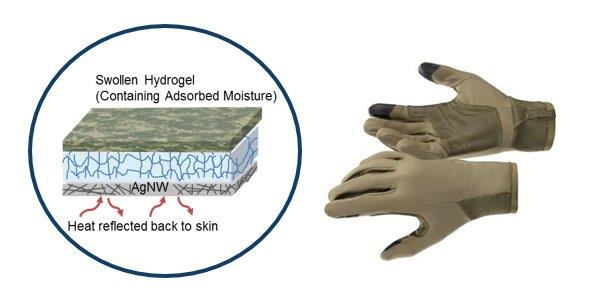
The work was inspired by research led by Yi Cui, Ph.D., at Stanford University. Cui’s team synthesized very fine silver nanowires, and then placed a network of the wires on cotton. By applying power to the silver nanowires, the researchers could heat the fabric.
D’Angelo, Elizabeth Hirst, Ph.D., and colleagues at the U.S. Army Natick Soldier Research, Development & Engineering Center, are working to extend this silver nanowire idea to fabrics suitable for military uniforms, such as polyester and a cotton/nylon blend. The Army team found that applying a mere 3 volts to 1-inch by 1-inch test swatches of these fabrics — the output of a typical watch battery — raises the temperature by 100 degrees Fahrenheit in just one minute. If these experimental fabrics can ultimately be used in uniforms, soldiers could dial up or down the voltage to vary the amount of heat their uniforms produce to match weather conditions. The added heating means uniforms could be thinner and lighter, an advantage for soldiers who must carry heavy loads and walk long distances.
The scientists are also incorporating a layer of sweat-absorbing hydrogel particles made of polyethylene glycol or poly(N-isopropylacrylamide). The researchers think these particles might stop other layers in the fabrics from getting wet, which would keep soldiers more comfortable during missions. After they return to base, the soldiers could release the sweat from their uniforms by hanging them up to dry in the warmer indoor air.
The silver nanowires on the fabrics can withstand repeated laundering, and now the researchers are working out how best to apply the hydrogel so it’s equally durable. D’Angelo and coworkers will also investigate how the silver mesh and hydrogel interact with each other. Another research direction for the team is to consider different power sources for the silver mesh, because batteries would add too much weight to uniforms.
Once D’Angelo and her colleagues optimize the fabric for gloves, they might extend the technology to clothing for the chest and legs. And ultimately, she says, it could make its way into consumer products.
The project is being supported by funding for basic research provided by the U.S. Army.
The American Chemical Society, the world’s largest scientific society, is a not-for-profit organization chartered by the U.S. Congress. ACS is a global leader in providing access to chemistry-related information and research through its multiple databases, peer-reviewed journals and scientific conferences. ACS does not conduct research, but publishes and publicizes peer-reviewed scientific studies. Its main offices are in Washington, D.C., and Columbus, Ohio.
Media Contact
ACS Newsroom
newsroom@acs.org