
There is no question about it, most of us crave sweet foods. In fact, many species of animals are the same way. No doubt it is due to our evolutionary history of seeking foods that had the greatest amount of energy to sustain us. Sugar has a lot of energy to go along with the sweet taste.
In order to maintain a healthy diet, health experts agree that sugar consumption should be limited. The effects of excessive sugar consumption can include higher blood pressure, inflammation, weight gain, diabetes, and fatty liver disease, which are all linked to an increased risk for heart attack and stroke.
For much of human history this wasn’t much of a problem, because there were few sources of pure sugar and most of the natural, sweet flavors came from honey, fruits, and berries.
When processed sugar became available, it was very expensive and out of reach for most people. But over time, as the sweetener became less expensive to produce, Americans began consuming more and more sugar.
The history and chemistry behind sugar production, and its impacts are a big part of U.S. history.
A Sweet History?
Explorer Christopher Columbus introduced sugar cane to the island of Hispaniola, in what is now the Dominican Republic, from the Spanish Canary Islands in 1493. Over the course of many years, cultivation of sugar cane spread throughout the Caribbean. The plant also thrived in parts of the U.S. states of Louisiana and Florida, where rich soil and warm temperatures were ideal for growing it.
But the process of growing and refining the sugar was extremely labor intensive and dangerous. When the first slave ships arrived in the New World in 1505, they began a system of forced labor to carry out the backbreaking work of sugar production. Over the next 300 years, some 10 million Africans were forcibly removed and enslaved to work on sugar plantations and other forced labor jobs.
The ancient process of making sugar was to take the canes, which were harvested in the fall, and send them to the mill to be pressed to release sugar cane juice. This juice was a dilute solution of sugar and water and it needed to be concentrated by evaporating the water. This was done over a series of large open kettles called a sugar train.
The kettles started with a very large size and worked down to successively smaller ones. The sugar cane juice was brought to near boiling in the first kettle, which could hold up to 500 gallons of juice. When the juice was at the proper thickness and quality, it was transferred to the next kettle. As water boils off and the solution becomes more concentrated, the boiling point of the solution increases due to a phenomenon known as boiling point elevation. The greater the concentration of dissolved solute, the higher the boiling point of the solvent. So, as the kettles got smaller and the sugar solutions more concentrated, higher temperatures were needed to continue the evaporation process.
When the sugar solution reached the smallest kettle, the sugar-maker would watch carefully. When it was at the perfect stage, they would strike it and pour the contents into cooling vats, where it would crystallize into pure sugar granules.
This process was both tricky and dangerous. Ladling the liquid from one vat to the next was hazardous work, and it was easy to get burned. The fire under each kettle had to be maintained constantly and even with all this work, the final step could fail and result in less valuable molasses instead of crystalline sugar.
But, in 1843 a chemical engineer came up with an invention that transformed the process completely and revolutionized the industry.

Boiling Points
The change between liquid and gas, otherwise known as evaporation, occurs at different temperatures and pressures depending on the liquid or solution.
For example, when water reaches 100 °C, the vapor pressure of the liquid equals the air pressure at sea level. This amount of heat energy gives the molecules of H2O at the liquid’s surface enough kinetic energy to overcome the hydrogen bonds that attract water molecules to each other.
Water molecules at the surface break free from surrounding molecules and escape as vapor into the space above the liquid. The vapor pressure is the pressure exerted by the gas in equilibrium with a solid or liquid in a closed container at a given temperature.
While in a closed system at a given temperature, some fraction of the molecules in liquids, or solids, have enough kinetic energy to overcome the intermolecular forces holding the particles together.
Those molecules break free from the surface, evaporating into gas. Simultaneously, some gas molecules condense back into the liquid or solid state at the same rate when the container is in equilibrium.
As a liquid is heated, the rate of evaporation and the vapor pressure increases. In an open container, when the vapor pressure is equal to the pressure of the gas surrounding the substance, it begins to boil.
This temperature and pressure is known as the boiling point. While vapor pressure is measured in a closed container, the air pressure of open containers affects the boiling point of liquids.
For example, at the peak of Mount Everest, it would take much longer to cook a pan of pasta. The mountain is more than 8,800 meters (29,000 feet) and, at that height, there are fewer air molecules pushing down on the surface of the liquid.
So, you only need to heat water to 68 °C (154 °F) at the top of the peak for its vapor pressure to equal that of the air pressure around it and for the water to boil.

The Multiple-Effect Evaporator Under Vaccum
Norbert Rillieux was born in New Orleans, La., in 1806, to a White father and Black mother. This meant that his status in Louisiana was uncertain. Though he wasn’t enslaved, Rillieux wasn’t awarded the same rights as whites in New Orleans.

So, Rillieux sought opportunity elsewhere, traveling to France to study engineering at École Centrale des Arts et Manufactures, a university in Paris. Recognizing his ingenuity, the university hired him as an instructor of applied mechanics at the age of 24.
Here, Rillieux studied steam engines, which had replaced animal and human labor as a source of power in factories. Alongside this research, Rillieux investigated the benefits of using the heat from steam to make the process of processing sugar cane more efficient.
As a result of his studies, Rillieux manufactured a device that was designed to evaporate sugarcane juice more efficiently and effectively: the multiple-effect evaporator.
In Rillieux’s invention, steam replaced wood heat to boil the sugarcane juice. The steam was produced by a single wood-fired boiler and then passed along to a series of closed evaporating pans, which replaced the open kettles. This made the heating process simpler and more efficient.
Another big innovation was to capture the heat coming off the boiling sugarcane juice and use it to heat the next evaporating pan down the line to get the maximum amount of work from the energy used to boil the sugar water. Previously, this heat was lost to the air as it came off the open kettles.
The last improvement was to reduce the amount of energy needed to evaporate the water. Rillieux did this by using pumps to reduce the pressure of the gas inside the evaporating pans. The boiling point is the temperature and pressure at which a liquid changes phase from a liquid to a gas. In everyday experience, we tend to ignore the role of pressure in this process. If you ask most people what the boiling point of water is, they will say 100 °C (or 212 °F). But that is only true at normal atmospheric pressure of 1 atmosphere or 760 millimeters of mercury.
If you reduce the pressure on the liquid it makes it easier for the liquid to change to a gas, and the temperature required to boil it goes down. For example, in Denver, Colorado water boils at 92 °C, and in La Paz, Bolivia, water boils at 87.7 °C. If the pressure is low enough, water can boil at room temperature.
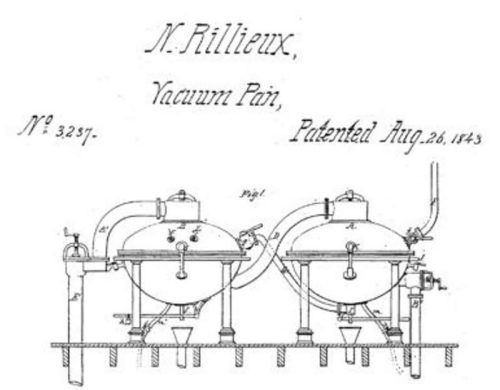
Since Rillieux was only interested in evaporating the water and not cooking the sugarcane juice, lowering the pressure was a great solution, saving time and energy. At the time of Rillieux’s invention in the 1830s, the laws of thermodynamics had not yet received widespread acceptance by the scientific community. The study of heat, temperature, energy, and matter, now known as thermodynamics, was a growing field while Rillieux was developing his multiple effect evaporator.
It wasn’t until around 1850 that Rudolf Clausius and William Thomson (Lord Kelvin) formally stated the first laws of thermodynamics. Like many other inventors, Rillieux came up with a means to apply the phenomenon he observed and fundamentally understood, long before scientists had a name to describe what was happening.
When Rillieux returned to the United States with his invention, he transformed the sugar-making process. When installing his multiple-effect evaporator on plantations where sugarcane was grown, however, he was denied residence in the houses of plantation owners, unlike his White counterparts, though his invention greatly improved their profits.
Additionally, at one point, his application for a patent was rejected because authorities assumed he was enslaved. Enslaved people did not have the rights of citizens, so Rillieux could not claim credit for his invention.
Because this process conserved fuel, an expensive cost for sugar producers, the technology was adopted around the world, including in Mexico, Cuba, Egypt, and France where sugar was refined. This early feat of chemical engineering was also applied to other processes such as the production of glue, soap, and condensed milk.
As the rights of free African Americans deteriorated prior to the Civil War, Rillieux left the United States to return to France. He never returned to the States. He died at 92 in his adopted country.
The Impact of Sugar on Health
The sweet stuff that made Rillieux famous was just one form of the broad category of food molecules known as carbohydrates. Saccharides is another name for sugars.
All carbohydrates are made of subunits called monosaccharides or simple sugars. Chemists classify carbohydrates based on the number of subunits in the compound. For example, monosaccharides have one sugar molecule, disaccharides have two, oligosaccharides have several, and polysaccharides have many.
Carbohydrates are found in virtually all foods, from apples to zucchini. These natural sugars in fruits and grain products are essential to sustain humans and provides energy to the body’s cells, tissues, and organs.
The sugar used as a sweetener in soft drinks, candy, and other sweets that once came from sugarcane is now more commonly made with high-fructose corn syrup produced from corn. How many food products that you regularly eat contain high-fructose corn syrup?
The added sugar may improve the taste and appearance of food, but as it has crept into diets around the world, the processed carbohydrates have worsened humans’ health. Teenagers who consume significant amounts of added sugar, often in the form of soda, have higher levels of “bad” cholesterol and triglyceride fats, which can negatively impact heart health later in life.
Higher levels of sugar consumption are linked to obesity, diabetes, and heart problems. About 0.35% of Americans under the age of 20 are estimated to have diagnosis of diabetes, a disease that prevents the body from either producing insulin or using it.
Insulin is essential to allow sugar from the blood to enter cells and be metabolized. 34.2 million people of all ages—or 10.5% of the U.S. population—have diabetes.
The rates of diabetes in the US population varies significantly among ethnic groups. The rate of diabetes in Native American and non-Hispanic Black adults is estimated to be almost double that of non-Hispanic Whites in the United States.
So, what constitutes a healthy diet? According to the U.S. Department of Health & Human Services publication, the Dietary Guidelines for Americans, a healthy eating plan:
- Emphasizes fruits, vegetables, whole grains, and fat-free or low-fat milk and milk products.
- Includes a variety of protein foods such as seafood, lean meats and poultry, eggs, legumes (beans and peas), soy products, nuts, and seeds.
- Is low in added sugars, sodium, saturated fats, trans fats, and cholesterol.
- Stays within your daily calorie needs.
The Nutrition Facts found on the back of food and beverages off ers a look into the amount of total and added sugars in each serving. The daily value provides a percentage of how much of these sugars the average person should consume. A good frame of reference is a daily value of 20% or above is a high source of added sugar and should be and should be consumed in moderation on a daily basis.
REFERENCES
Norbert Rillieux and the Multiple Effect Evaporator. Dillard University, New Orleans, La., National Historic Chemical Landmark, American Chemical Society, April 18, 2022: https://www.acs.org/content/acs/en/education/whatischemistry/landmarks/norbertrillieux.html [accessed Sept 2022].
Benfey, C. Degas in New Orleans: Encounters in the Creole World of Kate Chopin and George Washington Cable, University of California Press: Berkeley, Calif., 1997; pp.
Muhammad, K. G. The Sugar that Saturates the American Diet Has a Barbaric History as the ‘White Gold’ that Fueled Slavery. The New York Times Magazine, Aug 14, 2019: https://www.nytimes.com/interactive/2019/08/14/magazine/sugar-slave-trade-slavery.html [accessed Sept 2022].











