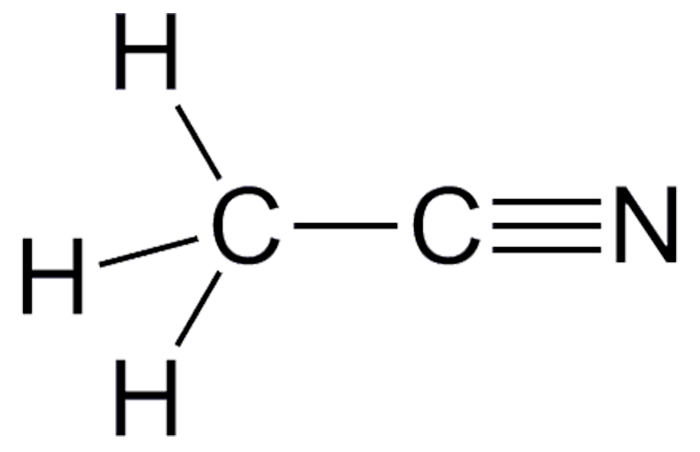
Acetonitrile is a solvent also known as methyl cyanide, cyanomethane, and ethanenitrile. It is colorless, volatile (bp 82 °C), flammable (flash point 2 °C), and toxic. It is miscible with water and most organic solvents except for some saturated hydrocarbons. J.-B. Dumas first prepared acetonitrile in 1847. In 1924, H. Adkins and B. H. Nissen developed the first acetonitrile manufacturing method by dehydrating acetamide.
Currently, acetonitrile is obtained as a coproduct of the acrylonitrile process. In 2008, the economic downturn decreased demand for acrylonitrile and reduced the supply of acetonitrile. To prevent future acetonitrile shortages, R. Fehrmann at the Technical University of Denmark (Kongens Lyngby) recently developed a “green” synthesis of the solvent from ethanol- and ammonia-derived ethylamine.

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

