What molecule am I?

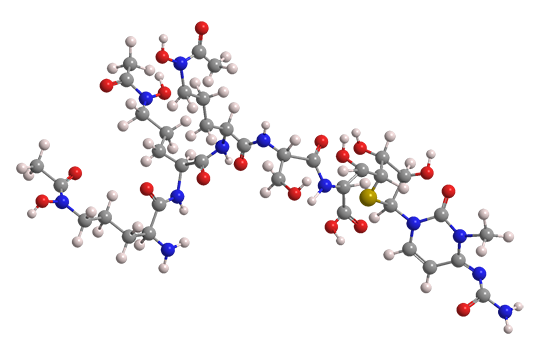
Albomycins are natural antibiotics produced by Gram-positive Actinomyces subtropicus bacteria. In nature, they exist as iron complexes. In the early 1950s, Russian biologist Georgii Frantsevich Gause was a pioneer in identifying albomycins and studying their antimicrobial properties.
The most abundant forms of albomycin are δ1, δ2, and ε (δ2 is shown), although at least three others exist. All albomycin structures are sequences of six modified amino acids. These antibiotics have shown activity against specific Gram-negative and Gram-positive pathogens.
Gause’s work with albomycins was largely forgotten until a few months ago. Natural products scientist Yun He and colleagues at Chongqing University (China) synthesized albomycins δ1, δ2, and ε and tested them in vitro against several strains of Streptococcus pneumoniae and Staphylococcus aureus, some of which are methicillin-resistant.
Albomycin δ2 was the most potent across the board, whereas the ε form showed no activity at all. Albomycin δ2’s potency exceeded that of the widely used drugs ciprofloxacin, vancomycin, and penicillin.
Albomycins are called “Trojan horse” compounds because bacteria sense that their iron-binding region is safe to ingest. Once inside, the sulfur-containing region (the “warhead”) inhibits the organism’s action.
Very little is known about the chemical properties and hazards of albomycin δ2, although its iron complex is highly soluble in water and therefore bioactive. Albomycin δ2 is difficult to synthesize and therefore commercially impractical, but simpler analogues that show similar activity are under investigation.
Albomycin δ2 fast facts
| CAS Reg. No. | 82113-67-5 |
| Empirical formula | C37H60N12O18S |
| Molar mass | 993.01 |
| Appearance | Yellowish solid |
| Melting point | Not determined |
| Water solubility | Not determined |

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

