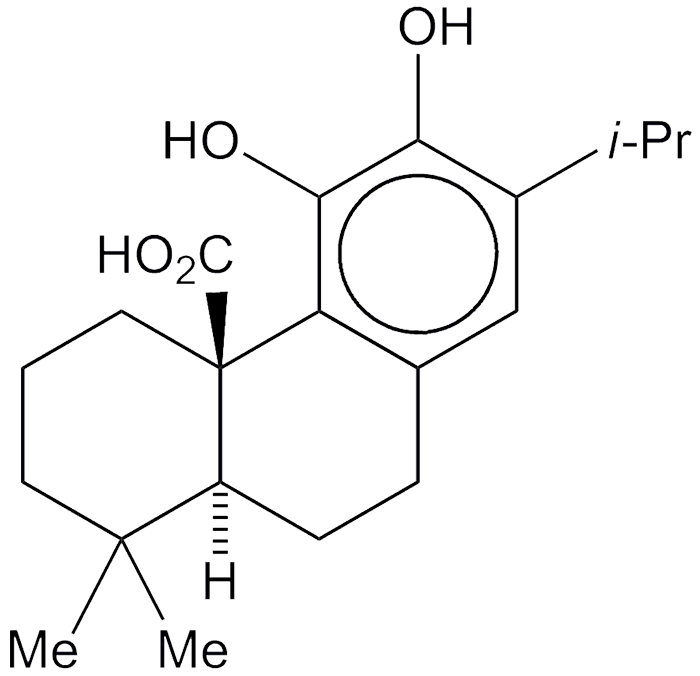
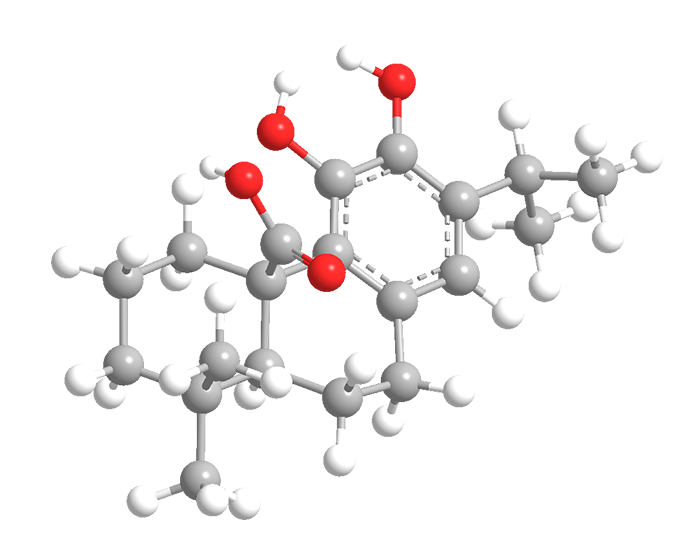
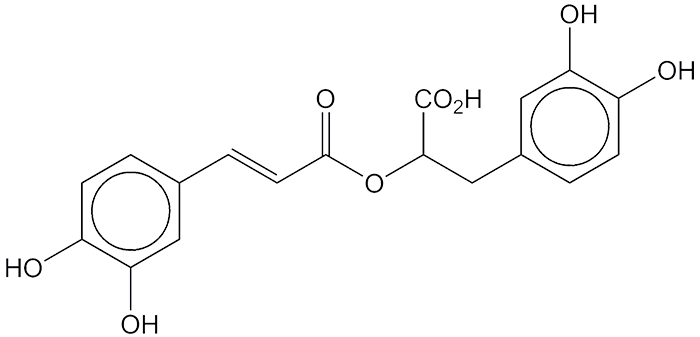
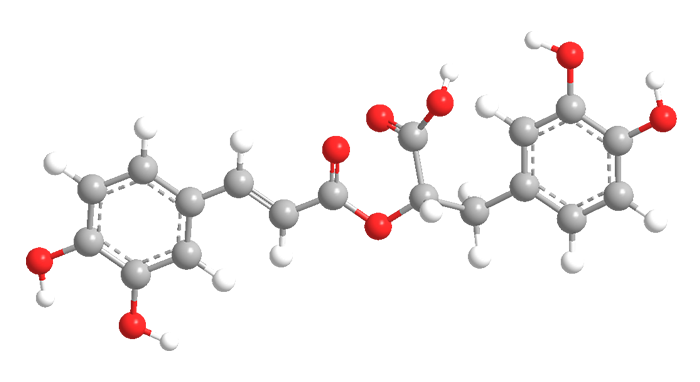
Carnosic acid (top two images) is a diterpene with an abietane skeleton; rosmarinic acid (bottom two) is a caffeic acid ester. Both are found in the rosemary plant (Rosmarinus officinalis), each also occurs in other plants.
But carnosic and rosmarinic acids have something else in common: They are meat preservatives. Rosemary extract was originally added to meat as a flavoring agent, but people noticed that it kept meat from spoiling. It was later discovered that the acids inhibit the free-radical reaction that oxidizes fats and oils and causes spoilage. Neither acid is responsible for the rosemary flavor.

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

