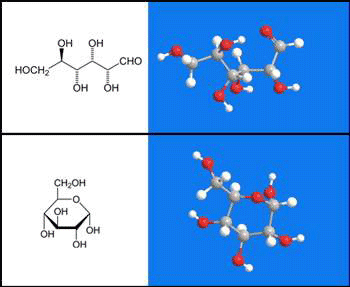
D-Glucose is the most common naturally occurring simple sugar and is a building block for disaccharides sucrose and lactose and higher oligo- and polysaccharides. It is the only sugar unit in cellulose and starch. Animals and plants produce D-glucose by glycogenolysis and photosynthesis, respectively, and use it as a primary energy source. D-Glucose is often depicted in the open form, but in solution more than 99% of it exists in one of two closed pyranose forms (α-D-glucopyranose is illustrated). Emil Fischer received the 1902 Nobel Prize in chemistry for his pioneering work in elucidating the structures of D-glucose and other sugars.
MOTW Update:
March 27, 2017
Last week, C&EN reported that a handheld glucose meter can be used to determine whether a patient is suffering from the flu or a bacterial infection. If the patient has the flu, viral neuraminidases in a nasal swab liberate galactose in a test strip. Another enzyme decomposes the galactose to glucose, which registers on the meter. If no glucose is detected, it is presumed that the infection is bacterial.

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

