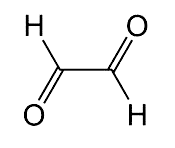
Glyoxal, the simplest dialdehyde, was first prepared by N. Lubawin in 1875, when he oxidized acetaldehyde with HNO3 or H2SeO3. The HNO3 method is still used commercially, along with metal-catalyzed gas-phase oxidation of ethylene glycol. At low temperatures, glyoxal forms yellow crystals; it melts at 15 °C and boils at 51 °C. Its low flash point (–4 °C) makes its green vapor explosive in air. It’s used chiefly in paper coating and textile finishing processes.

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

