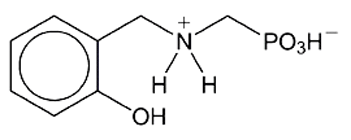
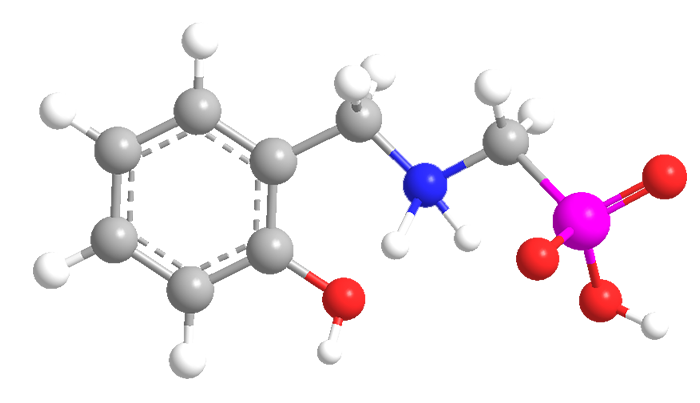
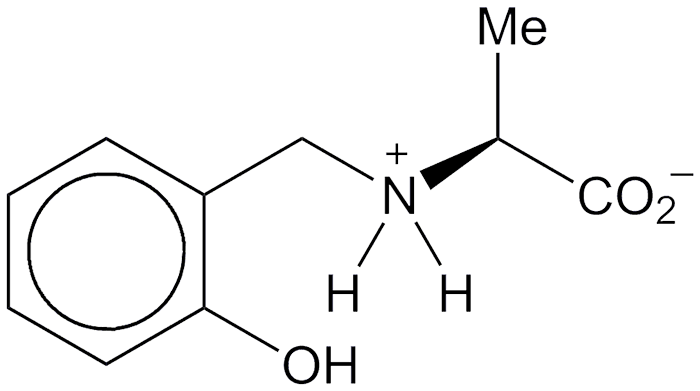
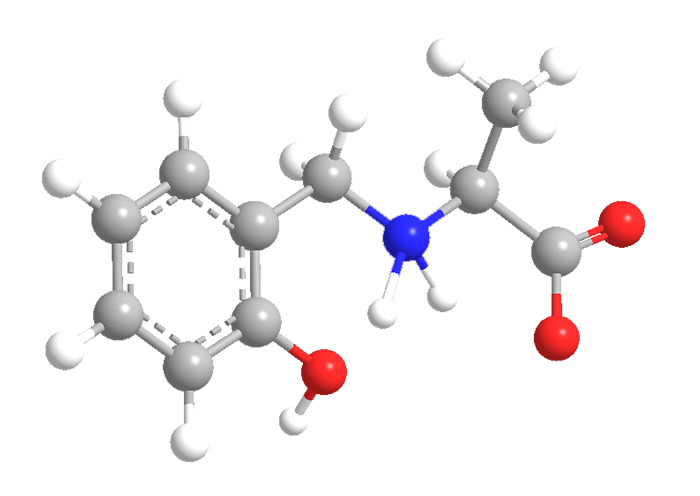
2-Hydroxybenzylalanine (HBA) and 2-hydroxybenzylaminomethylphosphonic acid (HBAMPA) are chelating ligands that have potential pharmaceutical applications. The molecules are analogues of each other: HBAMPA is a phosphonic acid, and HBA is the corresponding carboxylic acid with an additional methyl group. Both compounds normally exist as zwitterions.
In 2010, Y. Z. Hamada and W. R. Harris reported that both molecules are chelating ligands for several inorganic cations. At 25 ºC and pH 4.0, HBAMPA primarily forms protonated dinuclear complexes of formula M2L2H3 with aluminum(III) and gallium(III); M is the metal and L is the ligand. In both cases, other complexes are in equilibrium with the main one. HBAMPA also forms a 1:1 complex (FeL) with iron(III) along with its monoprotonated counterpart, FeLH. The complexes of the three metal ions differ sharply in their solubilities in the pH range 2–6.
HBA likewise forms complexes with Al(III) and Fe(III), although these complexes are less well characterized than those with HBAMPA. The iron complex precipitates from aqueous solution at pH >3.3, whereas the aluminum complex remains dissolved regardless of the pH level.

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

