What molecule am I?
The term “molecular motor” was introduced in the 1990s to describe protein “machines” that produce motion at the cellular level. At the same time, chemists initiated attempts to “build” molecules that, upon input of energy, performed work, typically rotating a portion of the molecule. The energy could be in the form of heat, light, electricity, or chemical reaction.
One of the first researchers to create molecular motors was Ben Feringa at the University of Groningen (The Netherlands). In 1999, he and his co-workers reported the synthesis of a photochemically driven molecular rotor. Last year, he and two other chemists were awarded the Nobel Prize in Chemistry for their pioneering work in this field.
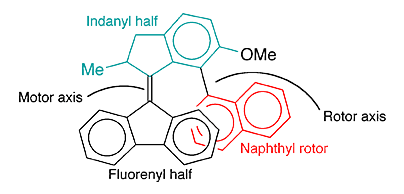
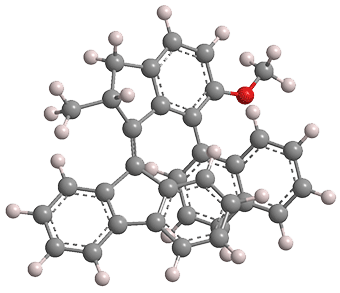
Earlier this year, Feringa and colleagues created a more complex machine: a molecular motor coupled to a rotor. The molecule, shown in the image, consists of a tricyclic fluorene group attached to a bicyclic indane via a double bond (the “motor axis”). The other ring of the indane is attached by a single bond (the “rotor axis”) to bicyclic naphthalene.
UV (365 nm) irradiation of the molecule at –60 ºC induces the double bond to isomerize and cause the indane–naphthalene moiety to rotate around the fluorene. The stereochemistry is such that only one side of the naphthalene ever faces the fluorene.
Feringa’s next objective is to “create machines that can amplify the molecular machines’ motion to larger movements or transmit motion over longer distances.”
MOTW Update
Salvinorin A, a hallucinogen obtained from the Mexican salvia plant, was Molecule of the Week for May 28, 2012. Recently, researchers synthesized an analogue of salvinorin A, 20-norsalvinorin A, that is easier to make and more stable than the natural product. The salvinorins show promise as anti-itching agents.

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

